Landy says, It makes the system gratuitously dependent on the proteins of the cell which serve as reporters of how well the cell is doing and where it is in its life cycle.
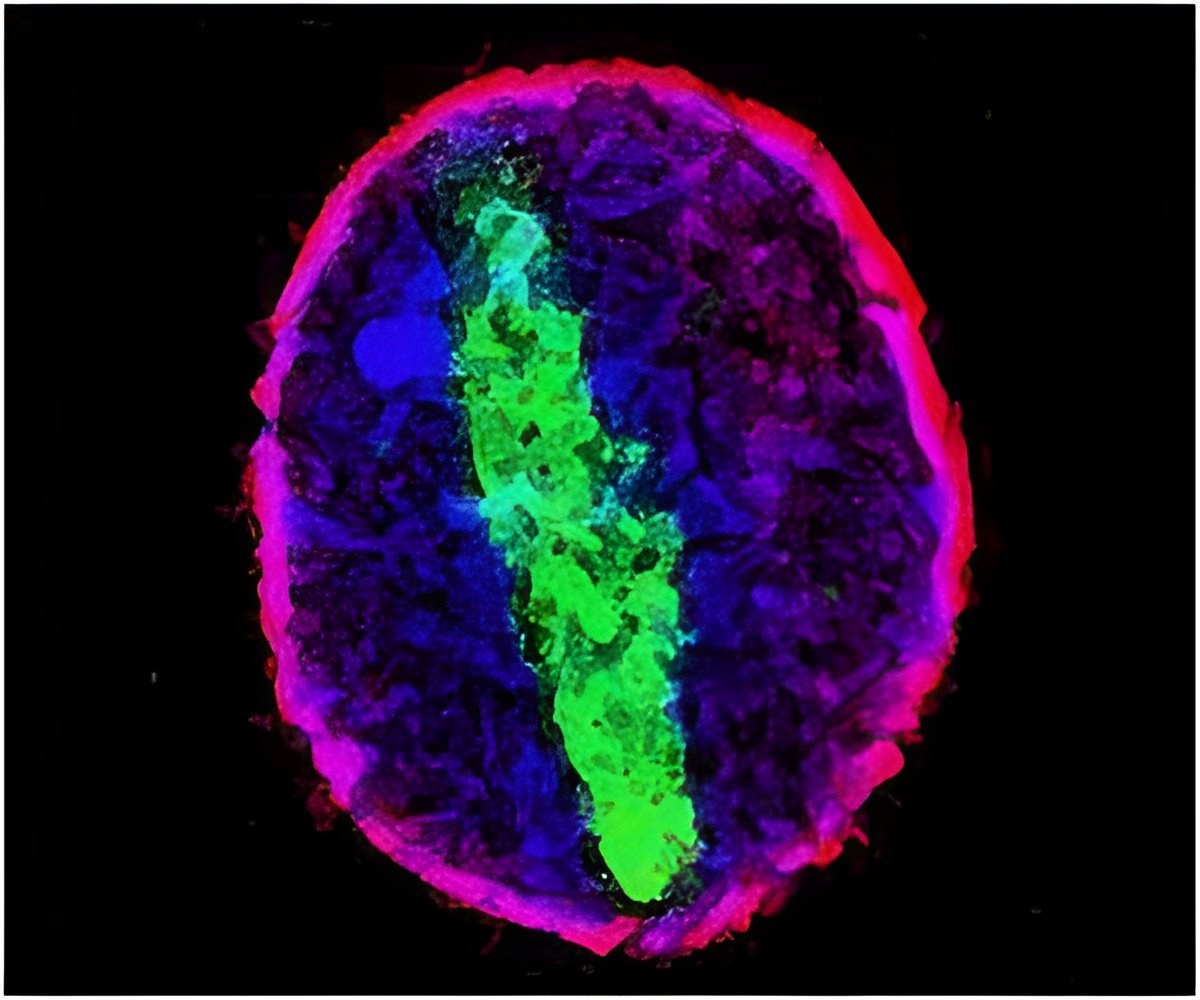
Mapping Bridges
Biologists already knew all this, but they had never quite figured out how the recombination proteins in the machines bridge the two DNA sites,(i.e.,which sites were bound or bridged by the same recombination protein). Therefore they couldn't really figure out what the whole machines looked like. Direct imaging tools such as crystallography never worked on the whole machines because it has too many forms, Landy said, and nuclear magnetic resonance never worked because it was too hard to make enough of the machines in a high enough concentration.
So the team did a couple of other experiments to figure out the bridging. One used chemicals and the other used genetics, but in each case they were essentially tagging different pairs of locations on the proteins and the DNA to see if any of those pairs, upon being connected, would produce a tell-tale effect: in the case of the chemistry the tell-tale was a change in the structural properties of the complex; in the case of the genetics it was a successful integration or excision reaction
It's a bit like trying to wire a battery into a light bulb circuit where there are many pairs of loose wires, but only two that actually connect to the bulb. When one finally connects (or "bridges") the right two loose wires with the battery's electrodes, the light bulb lights and then one has mapped where those correct two wires are.
They combined their new mappings, including distance measurements within the protein-DNA complexes using fluorescent tags, with all of the other biochemical and structural information biologists have learned over the last 50 years, into a computer model to render the overall depictions of the machines.
So now, long after they began to understand how worthwhile the Lambda virus was to study, researchers can finally see what these model DNA insertion and removal machines look like. That information will serve to make Lambda an even more powerful model for research and teaching, Landy said.
 MEDINDIA
MEDINDIA
 Email
Email




