Bristol-Myers Squibb and Pfizer have announced that federal regulators have expanded approval of their blood thinner Eliquis to treat DVT and PE
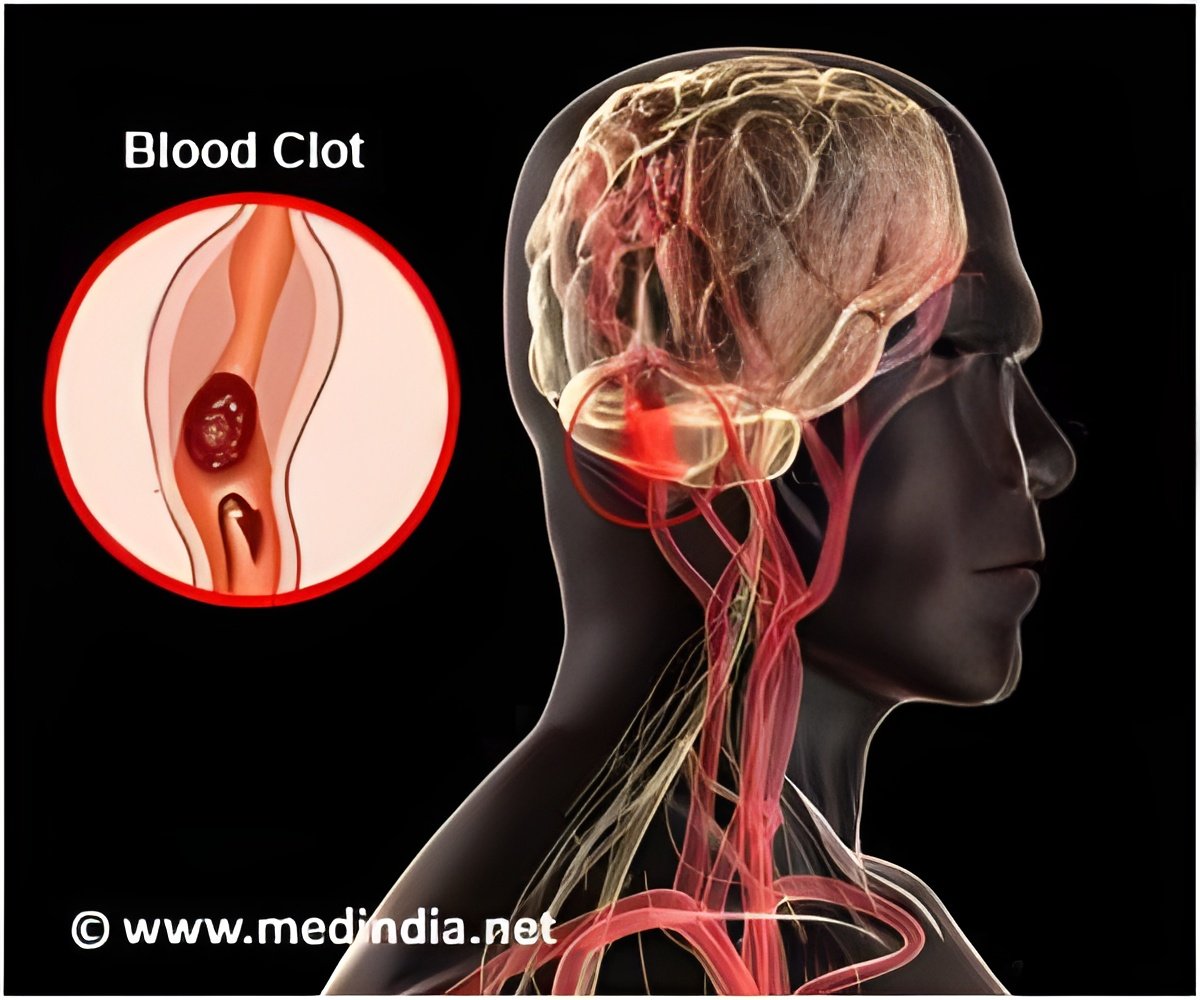
Eliquis, known chemically as apixaban, is manufactured by Bristol-Myers Squibb and co-marketed with Pfizer. Both companies are based in New York.
“We are pleased that Eliquis is now available as an effective treatment option for Deep Vein Thrombosis and Pulmonary Embolism. It offers oral dosing, no routine coagulation testing, and does not require the use of a parenteral anticoagulant or bridging during initiation,” said Douglas Manion, M.D., Head of Specialty Development, Bristol-Myers Squibb.
Source-Medindia
 MEDINDIA
MEDINDIA


 Email
Email










