University of Maryland School of Medicine has begun Ebola vaccine trails in Mali, said authorities.
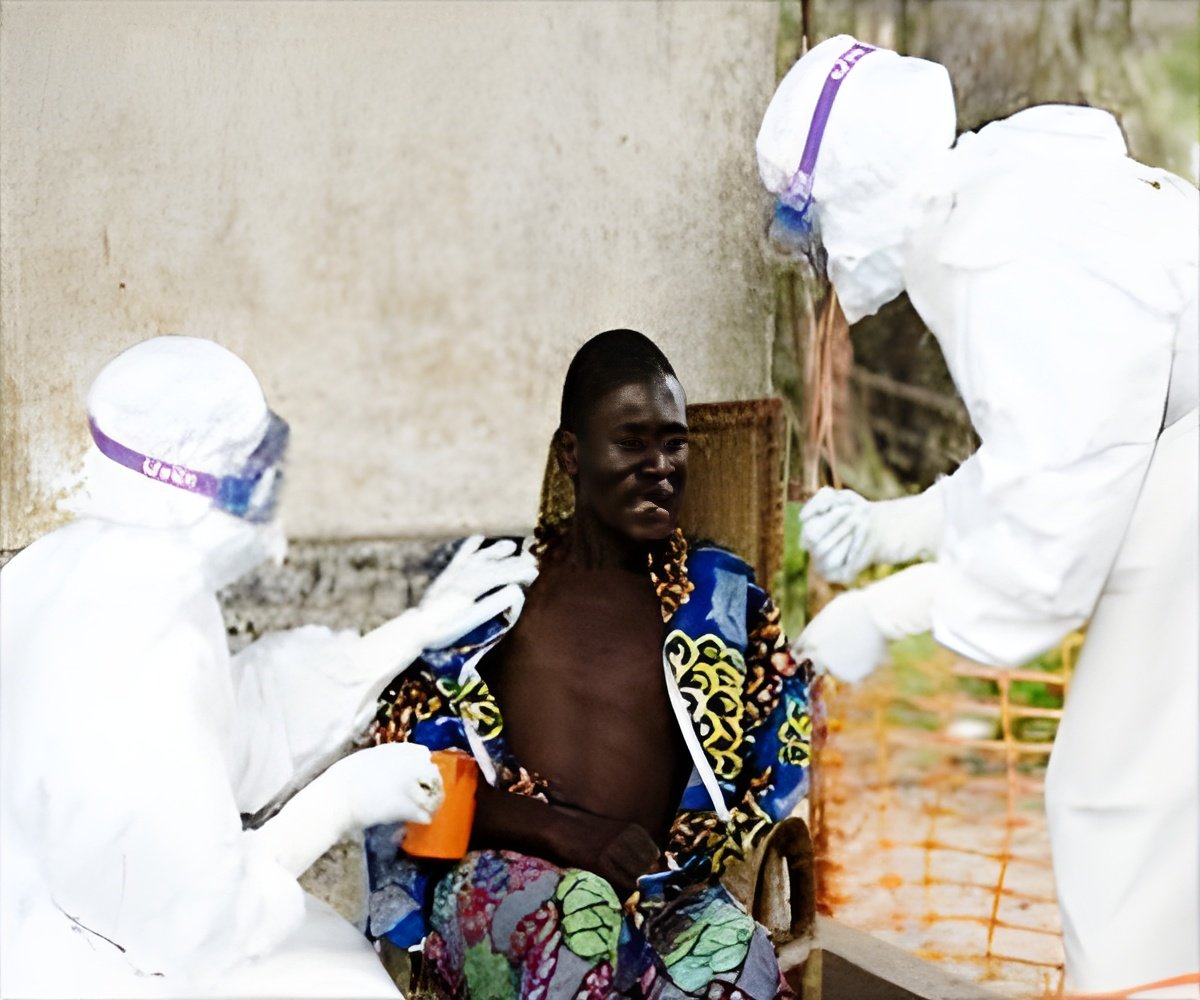
"Ebola is among the most urgent international public health issues we are facing. This research will play a key role in helping to solve it," said Dean E. Albert Reece, MD, PhD, MBA, as well as vice president of medical affairs, the University of Maryland and the John Z. and Akiko Bowers Distinguished Professor. "Dr. Levine, Dr. Sow and Dr. Tapia have done an extraordinary job, and are working hard to contribute their expertise to the worldwide effort to fight this virus."
"Pre-clinical research in primates by the VRC and Okairos, a biotechnology company acquired last year by GSK, indicate that the vaccine provides protection in non-human primates exposed to Ebola without significant side effects." The recent increase in funding for Ebola vaccine research is also enabling GSK to begin manufacturing at least 10,000 additional doses of the vaccine, even as the first clinical trials are occurring.
"This is impressive work by multiple groups to get this trial off the ground very quickly," said Dr. Levine. "It is a testament to everyone's commitment to fighting Ebola as aggressively as possible."
Source-Eurekalert
 MEDINDIA
MEDINDIA

 Email
Email








