Escherichia coli (E. coli) toxin can change surface of human intestine to benefit themselves, as per the team of researchers at Washington University School of Medicine in St. Louis.
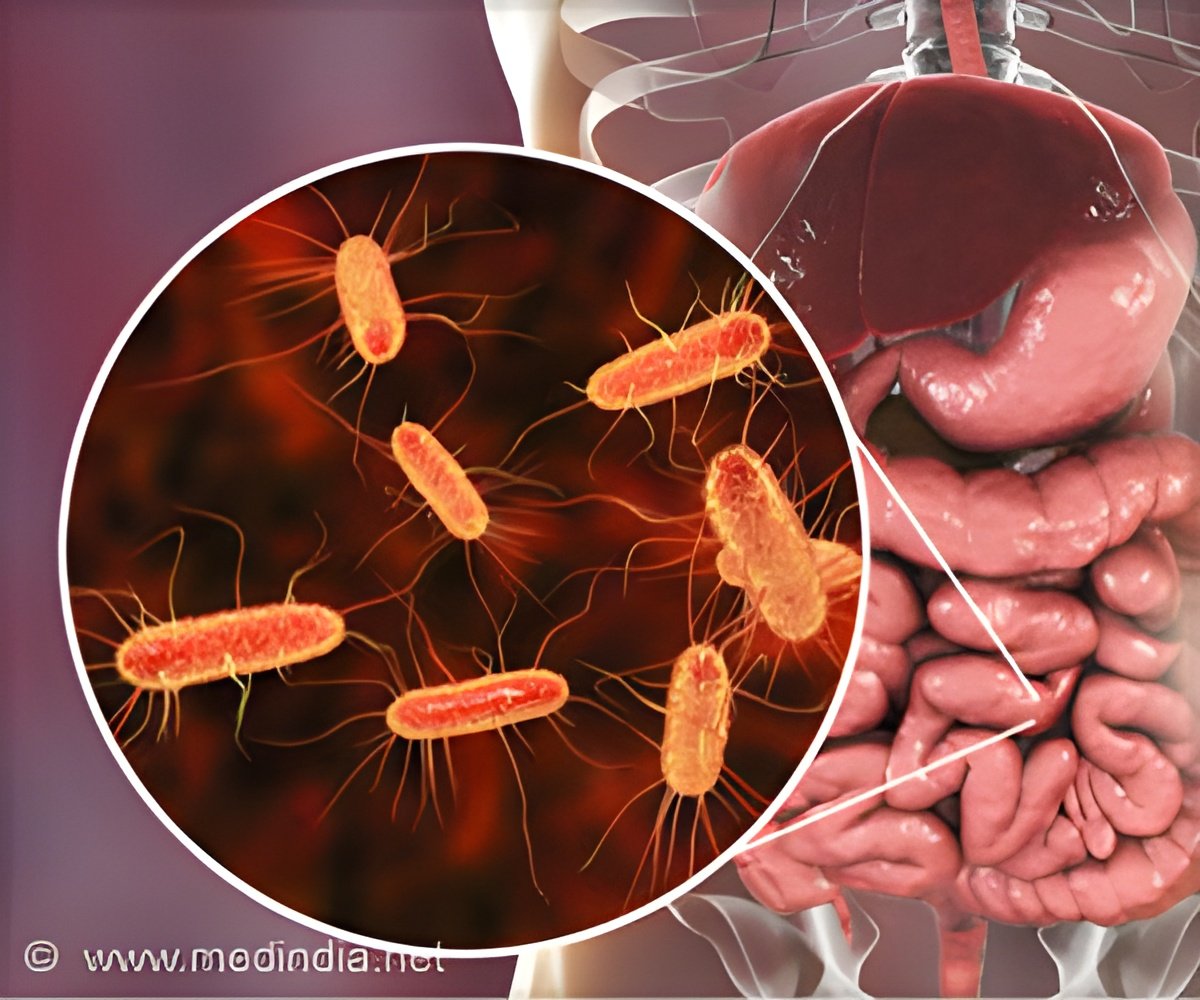
‘ETEC's heat-labile toxin is doing more than just causing acute diarrhea and dehydration. It is damaging the intestinal surface, that could affect the ability to absorb nutrients.’
Read More..




"There's more than meets the eye with this toxin," said senior author James M. Fleckenstein, MD, a professor of medicine and of molecular microbiology. "It is basically changing the surface of the intestine to benefit itself, probably ultimately to the detriment of the host.Read More..
Decades ago, people worked out how the toxin causes diarrhea, but until recently, nobody really had the tools to delve into what else this toxin might be doing. We're trying to put together the pieces of the puzzle to find out how toxin-producing E. coli might be driving malnutrition and other ripple effects of diarrhea."
Fleckenstein and first author Alaullah Sheikh, PhD, a postdoctoral researcher, study enterotoxigenic E. coli (ETEC), a toxin-producing strain of E. coli that is a common cause of severe, watery diarrhea. The bacterium's so-called heat-labile toxin causes ion channels on intestinal cells to open, triggering an outpouring of water and electrolytes into the digestive tract -- in other words, diarrhea.
Since oral rehydration therapy was invented in the 1970s, deaths from diarrhea have dropped by more than 80% worldwide. While invaluable at helping people survive a bout of diarrhea, the therapy does nothing to reduce the number of cases.
Worldwide, young children still develop diarrhea an average of three times a year, with the youngest and poorest children bearing the brunt of the caseload -- and of the long-term health consequences.
Advertisement
To find other ways the toxin affects the gut, the researchers grew human intestinal cells in a dish and treated the cells with the toxin. They found that the toxin activates a set of genes known as CEACAMs. One in particular -- CEACAM6 -- codes for a protein that is normally in cells of the small intestine at low levels.
Advertisement
"CEACAM6 is expressed in what is called the brush border of the small intestine, which is where all your vitamins and nutrients get absorbed," Sheikh said.
"This is one of the first pieces of evidence that ETEC can change the intestinal surface. We don't yet know how long that lasts and what that means for people who are infected, but it stands to reason that damage to this part of the body could affect the ability to absorb nutrients."
Fleckenstein, Sheikh and colleagues are continuing to study the link between ETEC and malnutrition, stunting and other health consequences.
"We are trying in the lab to understand the role of ETEC and its toxins as they relate to nondiarrheal effects of ETEC infection, particularly in young children in developing countries," Fleckenstein said. "There's a lot of work to be done to explore how the toxins might be related to these long-term consequences of diarrhea."
Source-Eurekalert