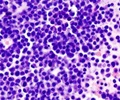
At Dartmouth-Hitchcock's Norris Cotton Cancer Center (NCCC), doctors have found a combination of drugs to potentially treat chronic lymphocytic leukemia (CLL) more effectively.
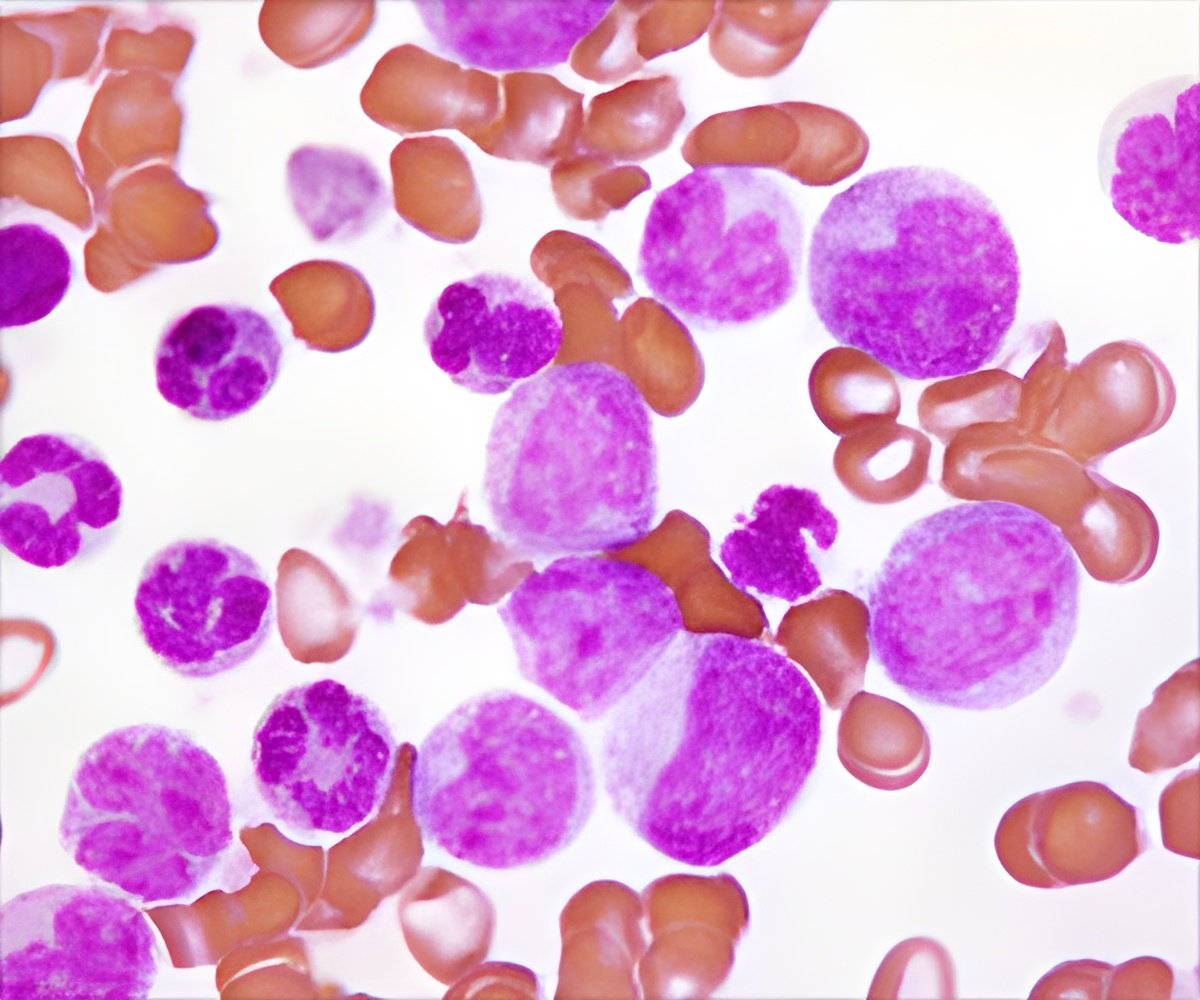
CLL lives both in the blood in circulation, and in lymph nodes and bone marrow. The former is relatively easy to kill, but the disease recurs because of resistant CLL cells in the lymph nodes and bone marrow. The researchers found an innovative drug combination that targets the stubborn CLL cells.
"We have been studying the mechanism in the cancer cells that causes the resistance to treatment," says Alan Eastman, the senior researcher on the team and a professor of pharmacology and toxicology at the Geisel School of Medicine at Dartmouth, "and that in turn, led us to find drugs that target the resistance." Eastman led the team at Dartmouth-Hitchcock's Norris Cotton Cancer Center that also included Ryan Soderquist, Darcy Bates, and Alexey Danilov.
The researchers found a very effective drug combination of gossypol plus navitoclax to kill CLL cells. "Both drugs have been given to patients, but never in combination, because no one had the mechanistic rationale for doing that. Now we have what we think is the most promising drug combination so far for the treatment of CLL," says Eastman.
CLL cells in the lymph nodes have an increased level of a protein known as BCL-X. Gossypol likely inhibits this protein, which allows the navitoclax to work more effectively to kill the cancer cells. Eastman and his team tested this drug combination on CLL immediately after they came out of the patients.
The willingness of the patients to participate was integral to this study," Eastman adds. "After 40 years of research, I think this is the most promising idea I have had that might truly impact patient outcome." The researchers hope a clinical trial will soon follow.
 MEDINDIA
MEDINDIA
 Email
Email