The claims of a top-selling drug have been questioned after a new research found that Lipitor does not reduce the risk of cardiovascular problems in women.
New research has found that Lipitor does not reduce the risk of cardiovascular problems in women leading experts to question the benefits of the drug in women.
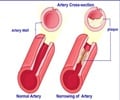
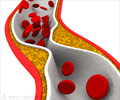
Lipitor has been the top-selling drug in the world and has accounted for over 12 billion dollars in annual sales.It has been prescribed to both men and women to lower cholesterol and reduce the risk of heart attack and stroke in patients with common risk factors for heart disease.
Now, the latest study was unable to find high quality clinical evidence documenting reduced heart attack risk for women in a primary prevention context.
Also, the study by Theodore Eisenberg of Cornell Law School and Martin T. Wells of Cornell University, also claims that the advertisements for the drug doesn't include label information relevant to women.
For the study, the researchers assembled studies for a meta analysis of drugs' effects on cardiovascular risk, considering all relevant studies reporting risks for both men and women.
No study including women with a mixture of risk factors for heart attacks gave any heed to the prescription of Lipitor or other statins to protect against cardiovascular problems.
Also, Lipitor's advertising repeatedly fails to report that clinical trials were statistically significant for men but not for women. Thus, unqualified advertising claims of protection against heart attacks may be misleading.
"Our findings indicate that each year, reasonably healthy women spend billions of dollars on drugs in the hope of preventing heart attacks but that scientific evidence supporting their hope does not exist," concluded the authors.
The study is appearing in the Journal of Empirical Legal Studies.
Source-ANI
RAS/SK
 MEDINDIA
MEDINDIA

 Email
Email





