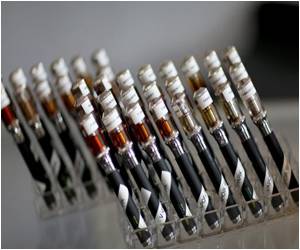
The move by the US regulators fell woefully short after the first restrictions on the soaring $2 billion market in e-cigarettes as stated by lawmakers and anti-tobacco advocates.

E-cigarettes are battery-operated devices that deliver vaporized nicotine into an aerosol inhaled by the user.
"This proposed rule is the latest step in our efforts to make the next generation tobacco-free," said Health and Human Services Secretary Kathleen Sebelius.
The new rules would bring e-cigarettes under many of the same rules that already apply to traditional cigarettes, including requiring sellers to enforce a minimum age restriction on those who wish to buy the products.
But the proposals from the US Food and Drug Administration (FDA) would not restrict advertising of e-cigarettes, nor would they ban the special flavors, such as Cherry Crush or Chocolate Treat, that some say are designed to appeal to children.
These gaps prompted strong criticism from lawmakers.
More is needed "to prevent e-cigarette companies from continuing to deploy marketing tactics aimed at luring children and teenagers into a candy-flavored nicotine addiction," said the group, which includes Senator Dick Durbin.
Waxman called the FDA proposals "an overdue but important first step."
The "FDA needs to act quickly to finalize this rule," he said, as well as "take additional steps to rein in manufacturers? marketing practices and use of candy flavors that target children and get them hooked on their addictive products."
- Health consequences? -
An industry group for e-cigarette makers, the Smoke Free Alternatives Trade Association, said it supported banning the sale of e-cigarettes to children.
But it objected to grouping e-cigarettes and related products with traditional cigarettes, arguing "they are technology products, not tobacco products," and therefore "deserve a new and distinct set of regulations."
But Roy Herbst, chief of medical oncology at Yale Cancer Center, said: "We do not know the nature of the long-term health consequences of these devices or what effect they will have on smoking continuation or uptake by adults and youth."
Herbst is chairman of a tobacco and cancer committee at the American Association for Cancer Research, which welcomed the FDA's proposed regulations.
The proposals include barring companies from handing out free samples and would required them to include health warning labels and to seek FDA approval before marketing a new product.
They would also have to register with the FDA and provide details about their ingredients.
And they would be prevented from advertising claims that they pose a lower health risk compared to traditional cigarettes, unless the FDA confirms scientific evidence backing up the claim.
E-cigarette use by young people has been booming: a December study by the US Centers for Disease Control and Prevention (CDC) estimated that 10 percent of high school students had used them.
In addition to the special flavors seen as targeting youth, most manufacturers have provided free samples at hundreds of events, including youth-oriented concerts, and broadcast TV or radio advertising.
The CDC has also reported a spike in calls to poison control centers about accidents involving the nicotine-filled bottles used to refill e-cigarettes.
More than half the calls involved children under five who had swallowed, inhaled or spilled the liquid on their skin or in their eyes.
The proposals will be open to public comment for 75 days.
Lawmakers as well as anti-tobacco groups are urging US regulators to finalize the rule as quickly as possible and put it into effect within the year.
Source-AFP
 MEDINDIA
MEDINDIA

 Email
Email