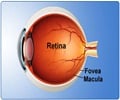
The adult mammalian central nervous system has only limited intrinsic capacity to regenerate connections after injury.

Many different approaches are being trialed, targeting different receptor systems and/or different signaling pathways, some aimed at enhancing intrinsic growth capacity in injured RGCs, others aimed at reducing the impact of factors external to the neuron that suppress/restrict the regenerative response. An approach increasingly of interest involves the use of modified, replication-deficient viral vectors to introduce appropriate genes into injured cells in the visual pathway (gene therapy).
In the perspective article written by Prof Alan Harvey, from School of Anatomy, Physiology and Human Biology, The University of Western Australia, he summarized recent gene therapy research from his laboratory, using the rodent visual system as an experimental model, which is aimed at improving both the viability and regenerative capacity of injured adult RGCs. These perspectives were published in the Neural Regeneration Research (Vol. 9, No. 3, 2014).
Source-Eurekalert
 MEDINDIA
MEDINDIA


 Email
Email