Cancer drug larotrectinib successfully targets cancers with tumor-specific gene mutations (tropomyosin receptor kinases - TRK) in adult and childhood cancers.
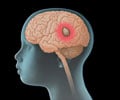
‘Larotrectinib, an oral drug that acts against proteins that contribute to tumor growth (tropomyosin receptor kinases - TRK), has durable antitumor activity in patients with TRK fusion–positive cancer, regardless of the age of the patient or the type of tumor. Tumors involving TRK occur in diverse childhood and adult cancers.’





"This drug represents a changing paradigm in cancer care where we evaluate a tumor, not only by where it exists in the body, but by the genetic mutations that are driving its growth," said Ramamoorthy Nagasubramanian, MD, an author of the study and division chief of pediatric hematology-oncology at Nemours Children's Hospital.The study integrates findings of three phases of research, including an adult phase 1, a pediatric phase 1/2, and an adolescent/adult phase 2 study, to report on the safety and efficacy of the drug. Fifty-five patients with TRK fusion-positive cancers, detected by molecular profiling as routinely performed by each site, were enrolled across the study sites. Patients ranged in age from 4 months to 76 years old and had 17 unique cancer diagnoses, including infantile fibrosarcoma, salivary gland tumors, and thyroid cancer. Each patient received two daily doses of the drug in pill or liquid form.
Overall 75 percent of patients had their tumors respond to the treatment, with 13 percent achieving a complete response and 62 percent achieving a partial response.
Responding patients remained on treatment or underwent surgery with curative intent. The median time to response was 1.8 months (range 0.9 to 6.4). The median duration of response and progression-free survival had not been reached, but after one year, 71 percent of patients with a response were ongoing and 55 percent of all patients remained progression-free.
The treatment was well-tolerated by patients. Clinically significant adverse events were uncommon and most frequently included inflammation in the liver and other organs (alanine or aspartate aminotransferase increase), fatigue, vomiting, and dizziness.
Advertisement
The study authors note that additional data reflecting longer follow-up and a larger patient population will provide further insight into the safety profile of the drug, as well as the durability of the response.
Advertisement
Source-Eurekalert