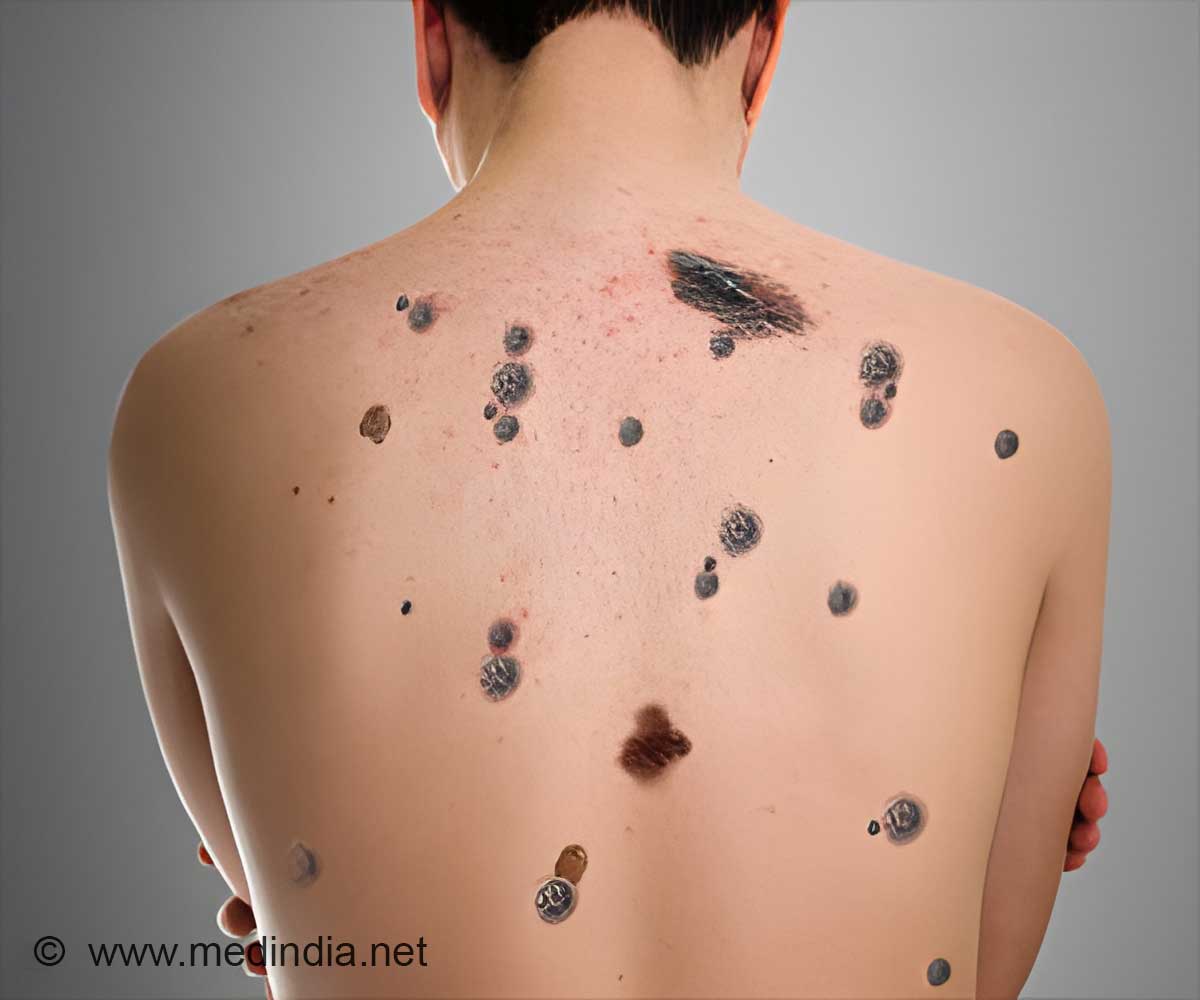
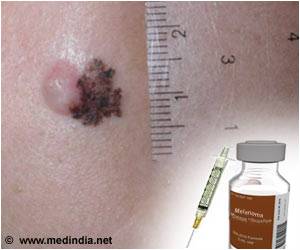
Pembrolizumab drug works by making cancer cells visible to the body's immune system so it can be attacked by its own natural defence mechanisms.
Gillian Nuttall, founder of Melanoma UK said, “We are delighted that patients with advanced melanoma will now be given another treatment option. Melanoma is a very difficult disease to treat and it is good news that this treatment will now be made available on the NHS. We must express our thanks to everyone who worked tirelessly in achieving this outcome. This is very good news for the melanoma community.”
The National Institute for Health and Care Excellence (NICE) approved guidelines for the use of the treatment, developed by Merck & Co Inc (MSD). It works by making cancer cells "visible" to the body's immune system so it can be attacked by its own natural defence mechanisms.
The NICE recommendation for pembrolizumab, is the first medicine available through the government Early Access to Medicines Scheme (EMAS), and is one of the fastest ever NICE appraisals for a treatment.
George Freeman, Life Sciences minister said, “I am delighted that MSD has decided to use the Early Access to Medicines Scheme to accelerate access for our NHS patients."
Mike Nally, UK Managing Director, MSD said, "We are delighted that NICE has been able to expedite its review of pembrolizumab and for taking the uncommon step of issuing a positive FAD recommendation following the first appraisal committee meeting.”
 MEDINDIA
MEDINDIA


 Email
Email








