Study discovers a mechanism that inhibits tumor growth in skin cancer by switching the immunosuppressive environment to an immunosupportive one.
TOP INSIGHT
If autophagy is blocked in tumor cells, they produce cytokines that attract natural killer cells.
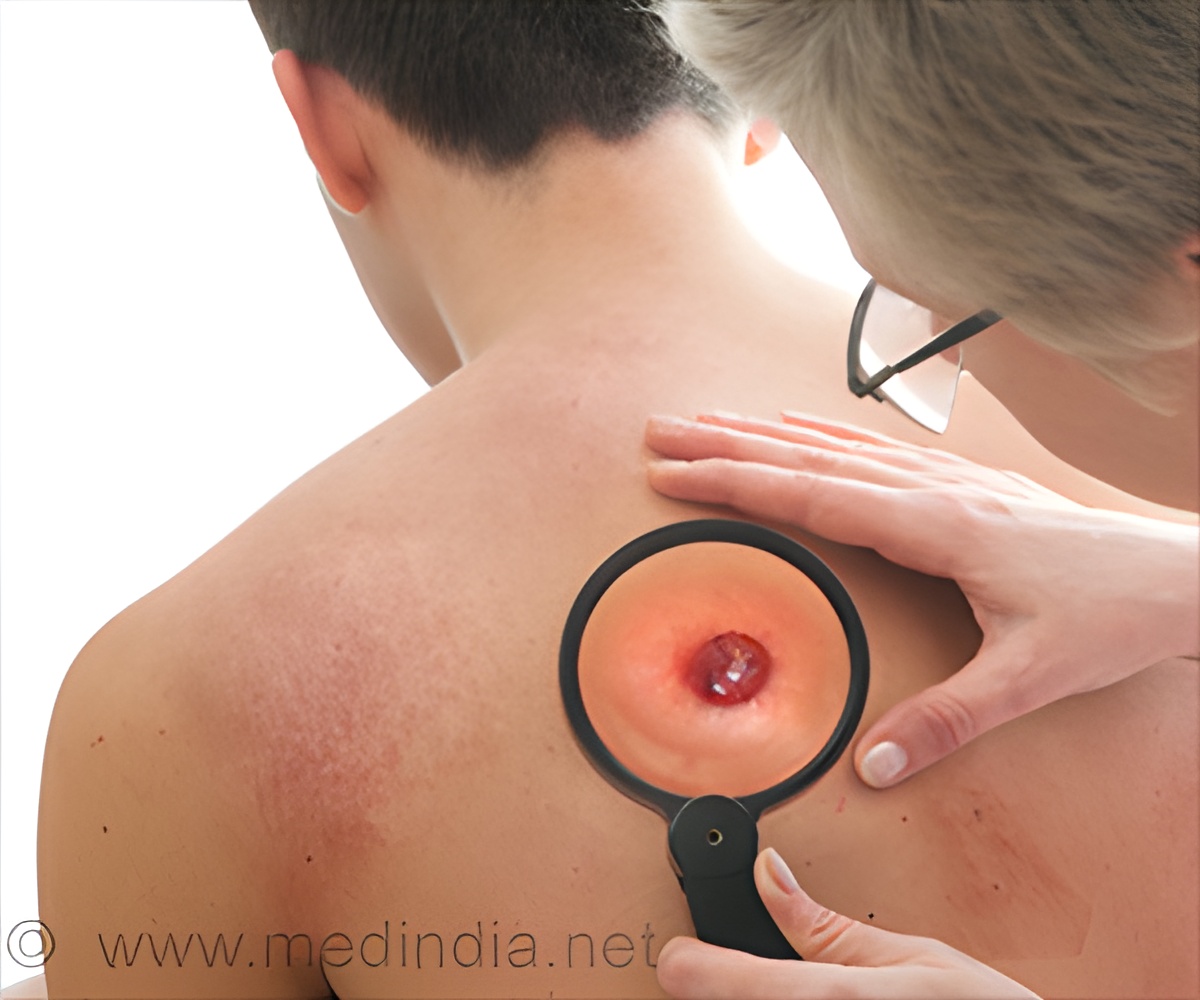
A critical hallmark of the malignant progression of tumors is evasion and suppression of the immune system. This occurs through the ability of tumor cells to develop an immunosuppressive microenvironment preventing cytotoxic immune cells such as NK cells to infiltrate tumors and kill cancer cells. Therefore, a key issue in the field of anti-cancer immunotherapy is to develop strategies capable of driving cytotoxic immune cells into the tumor bed.
For several years already, it is well known that tumor growth can be reduced by suppressing autophagy in cancer cells. While autophagy blockade is believed to render tumors more sensitive to chemotherapy, its impact on anti-tumor immunity is not well understood. Autophagy can be blocked in tumor cells and mouse models pharmacologically and genetically, the latter by inhibiting the expression of genes involved in the initiation of autophagy, for example BECN1 coding for the protein Beclin-1.
Targeting autophagy impacts on NK cells
The research team of Dr Bassam Janji in the Laboratory of Experimental Cancer Research at LIH's Department of Oncology is specialized in the study of the tumor microenvironment in different types of solid tumors, including skin cancer. In their newly published study, the researchers aimed to investigate the effect of blocking autophagy on the infiltration of NK cells in melanoma.
Using tumor biopsies from melanoma patients, the researchers could show that there is a positive correlation between the production of CCL5 and the infiltration of NK cells. The more CCL5 is produced, the stronger tumors are infiltrated with NK cells. A high level of production of CCL5 was also found to have a positive impact on the survival of melanoma patients.
This is the first time that a mechanistic link between autophagy and NK cell recruitment could be established. Dr Janji sees a high therapeutic potential in the discovery. "Targeting autophagy in tumor cells is a promising strategy to reinforce the immune system to fight cancer", he claims. "Our study provides a cutting edge advance in the field of cancer immunotherapy and could specifically pave the way for more effective NK cell-based immunotherapies. We now know how we can switch an immunosuppressive tumor environment to an immunosupportive one, enabling the action of NK cells. NK cells have a strong anti-tumor potential, but it can only be exploited if the cells are efficiently directed to the tumor. "
The researchers are now planning to address the impact of targeting autophagy either genetically and pharmacologically on the immune landscape of melanomas by phenotyping other immune cells in the melanoma tumor microenvironment. The ultimate goal of Dr Janji's research team is to provide the proof of concept that targeting autophagy could improve the efficacy of current immunotherapies, notably those based on immune checkpoint blockades.
Source-Eurekalert
 MEDINDIA
MEDINDIA
 Email
Email






