A new device developed by University of Cincinnati researchers could appear as a blessing for women who experience severe menstrual pain.
A new device developed by University of Cincinnati researchers could appear as a blessing for women who experience severe menstrual pain.
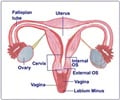
Fed up of the chronic pain, women often end up having medications that can cause gastrointestinal side effects.Clinical trials have shown that the new vaginal device safely delivers at least 10-times more drug to the uterus as a tablet of equivalent dose.
During the study, the researchers recruited 18 participants, aged 18-45 years with menstrual cycles between 25-30 days.
In the mid-follicular phase of the first menstrual cycle (days 7-11), nine study participants received an oral dose of 10 mg of ketorolac (Toradol), a non-steroidal anti-inflammatory medication; while nine women received a tampon coated with 10 mg of ketorolac.
During the second menstrual cycle, each subject received the opposite treatment.
The results revealed that the medication administered vaginally does not cause significant side effects but accumulates more efficiently in the desired uterine tissue than using the oral medication.
"Phase II clinical trials will study efficacy of the treatment to assess whether the drug concentration is effective in reducing pain," Pauletti added.
Source-ANI
ARU
 MEDINDIA
MEDINDIA



 Email
Email