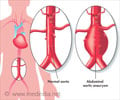
HEAL, the new personal health management tool may one day help diagnose, treat, and prevent diseases such as abdominal aortic aneurysm.

TOP INSIGHT
HEAL - the new personal health management tool may one day help diagnose, treat, and prevent diseases such as abdominal aortic aneurysm, HEAL can also detect neurological diseases such as autism and schizophrenia as well as cardiovascular diseases and chronic diseases of aging.
"Our study presents a new framework for disease genome analysis," he says. Disease outcomes usually result from the combined effect of personal genomes and lifestyle, making it difficult to diagnose certain diseases early or predict an individual's prognosis, says co-author Michael Snyder, director of genomics and personalized medicine at Stanford. HEAL's integrative model identifies genome baselines for a given disease and provides actionable guidelines, he says.
HEAL may one day allow physicians or genetic specialists to confer remotely with patients about the patient's personal genetic makeup. "In the long run, the patient would not visit the office at all unless they need to," Snyder says. "They could mail in their saliva sample, get back their genome sequence, and get return of results through video teleconference."
HEAL would provide the physician with a "risk score" encompassing recommendations about treatment and lifestyle. Snyder cautioned that the risk scores do not mean that a person will definitely get a particular disease. "We need to educate both the physician and patient about that," he says.
Using machine learning, in which computers extrapolate results from data on their own, HEAL processed whole-genome sequencing data, physiological measurements from patients' clinical visits, and their smoking histories. The system analyzed 313 individuals with AAA and 161 control cases to identify 60 genes important for AAA. The results were later confirmed in human aortic tissues and mouse models.
While widespread genome profiling holds tremendous promise in predicting and managing disease, sequencing the DNA of every individual at birth or later in life raises privacy concerns. "The genome data belongs to the person and it is up to them to decide how to share it," Snyder says. "If placed in a medical database, it should be secure."
 MEDINDIA
MEDINDIA



 Email
Email









