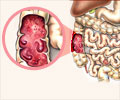
A new study in mouse model has shown that infusions of a particular bone marrow stem cell appears to control inflammatory bowel disease.
Infusions of a particular bone marrow stem cell appears to control inflammatory bowel disease as evidenced by a new study on mice.
Researchers at the Massachusetts General Hospital (MGH) found that mesenchymal stem cells (MSCs), known to control several immune system activities, allowed the regeneration of the gastrointestinal lining in mice with a genetic mutation leading to multiorgan autoimmune disease."Our findings suggest that MSC therapy could become a useful treatment for inflammatory bowel disease," said lead author Biju Parekkadan, PhD, of MGH Center for Engineering in Medicine.
"Several previous studies have observed these cells' ability to inhibit specific subsets of T cells and relieve symptoms in particular autoimmune disorders. But this is the first demonstration of their ability to suppress a broad-based autoimmune reaction and protect gastrointestinal tissue," Parekkadan added.
Found in the bone marrow, MSCs give rise to tissues supporting blood cell development and secrete factors that can modulate several immune system activities. Their use has recently received FDA approval to treat severe graft-versus-host disease in children.
The new study was designed to find MSCs' therapeutic potential in a model of multiorgan autoimmune disease.
The researchers used a strain of mice in which a genetic mutation leads to deficiency in regulatory T cells, which suppress the activity of self-reactive immune cells, resulting in overwhelming autoimmune disease.
While little improvement was seen in the pancreatic or liver tissue, in four of the six MSC-treated mice, intestinal tissues appeared almost identical to those of normal mice.
Analysis of the animal's lymph nodes revealed that MSC treatment produced a significant reduction in inflammation.
Surprisingly, cell-tracking studies indicated that the MSCs - which were administered by infusion into the peritoneum, the membrane lining the abdominal cavity - moved into abdominal lymph nodes rather than to the intestine itself.
The presence of MSCs was associated with a reduction of activated T cells and changes in other indicators of immune system activity, indicating suppression of the out-of-control immune reaction.
The study is published in the journal Stem Cells.
Source-ANI
RAS/M
 MEDINDIA
MEDINDIA


 Email
Email