The European Medicines Agency has recommended the approval of oral Ketoconazole HRA for the treatment of Cushing’s syndrome.

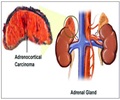
Cushing’s syndrome is a hormonal disorder where there is excess of the hormone cortisol in the body. The condition can be life-threating, with the patient suffering from diabetes, hypertension and other complications. Cortisol is normally produced by the adrenal glands, and its secretion is controlled by parts of the brain called the hypothalamus and pituitary. Thus, a pathology like a tumor in any of these parts could result in Cushing’s syndrome. In addition, Cushing’s syndrome is also caused by intake of high doses of glucocorticoids over prolonged periods. These patients may recover once their dosage is reduced. In instances where the Cushing’s syndrome is due to a tumor, surgery or radiotherapy may be necessary.
Ketoconazole:
Ketoconazole is an antifungal drug that is commonly used as a topical medication against fungal infections. Its oral form is used very cautiously or is banned in some countries due to its damaging effects on the liver. Beside its antifungal effects, ketoconazole is also an enzyme inhibitor and has to be used cautiously with other medications.
Ketoconazole HRA for Cushing’s syndrome:
The European Medicines Agency has recommended the approval for use of Ketoconazole HRA for the treatment of Cushing’s syndrome. Ketoconazole is effective in the treatment of Cushing’s syndrome due to its enzyme inhibitory activity. Ketoconazole inhibits the enzymes necessary for the formation of cortisol hormone, and thereby reduces the level of the hormone. This approach could be especially useful in those individuals who cannot undergo surgery for some reason, or in whom surgery does not produce expected results. The beneficial effects of ketoconazole in Cushing’s syndrome are well documented, since it was being used for this purpose for several years despite not having an official approval.
As Ketoconazole HRA moves forward in the approval process for the treatment of Cushing’s syndrome, it is hoped that several people with Cushing’s syndrome will benefit from the use of this medication in the near future.
Source-Medindia
 MEDINDIA
MEDINDIA
 Email
Email