The National Institute for Health and Care Excellence (NICE) in UK has recommended the drug.
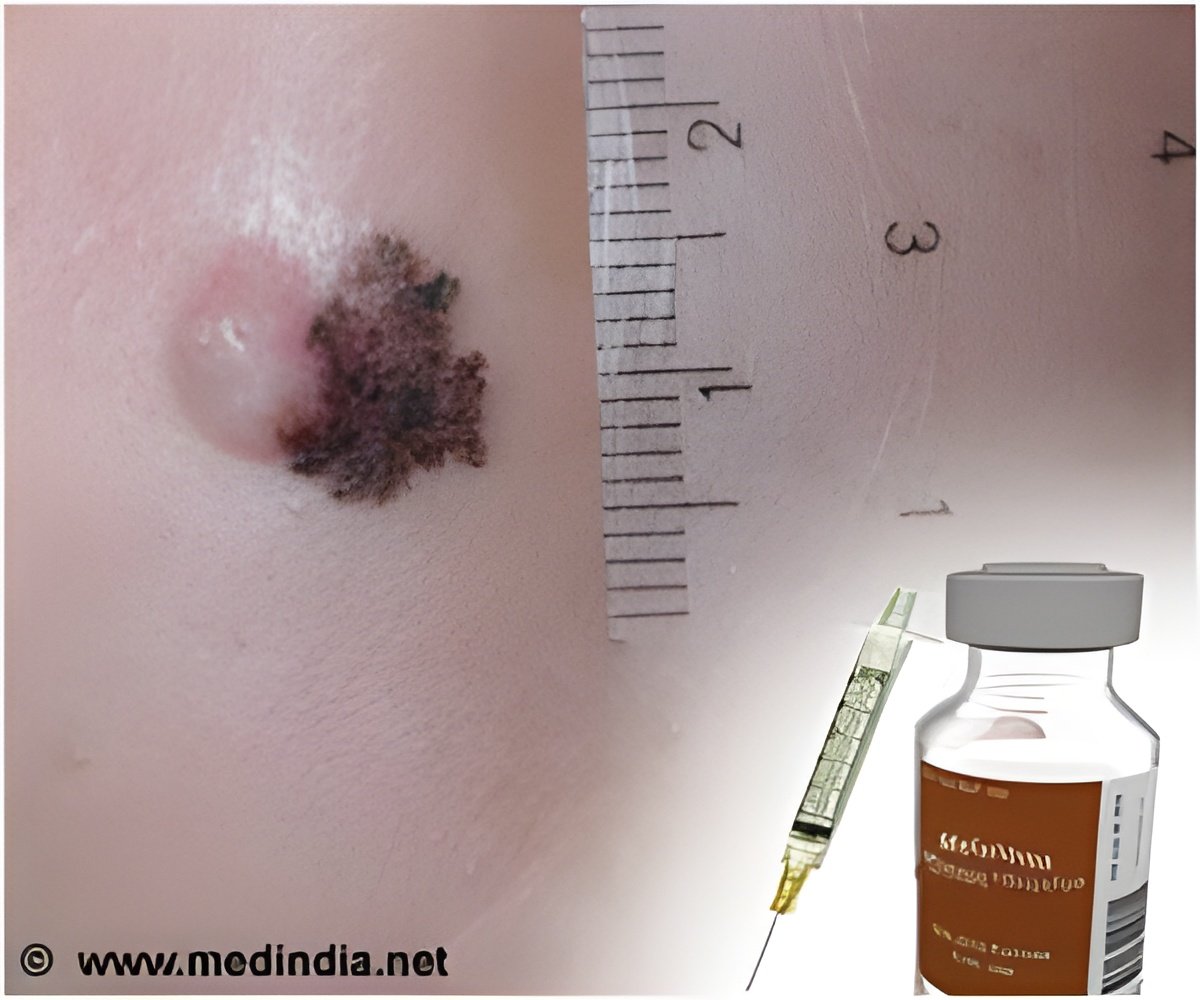
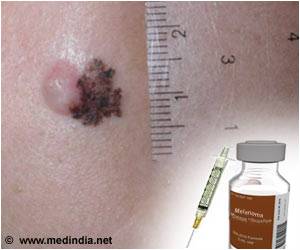
With GlaxoSmithKline agreeing to provide its melanoma pill Tafinlar at an undisclosed discount price, the National Institute for Health and Care Excellence (NICE) in UK has recommended the drug for treating unresectable or metastatic BRAF V600 mutation-positive melanoma.
Tafinlar is similar to Roche's Zelboraf and targets a genetic mutation known as BRAF that is linked with melanoma. Results from the BREAK-3 trial revealed that the drug was more effective than dacarbazine in treating patients with the specific kind of melanoma.
NICE’s recommendation of the drug has received approval from physicians in the UK. Speaking to the BBC, London's Institute of Cancer Research’s Paul Workman said, “Its approval underlines the importance of a new generation of cancer drugs targeted at specific molecular features of [tumors]--drugs which after years of painstaking development are now making their way to patients.”
Source-Medindia
 MEDINDIA
MEDINDIA

 Email
Email







