The first steps of origin of pancreatic cancer have been identified, claim researchers at Mayo Clinic's campus in Jacksonville. They added that the findings suggest preventive strategies to explore.
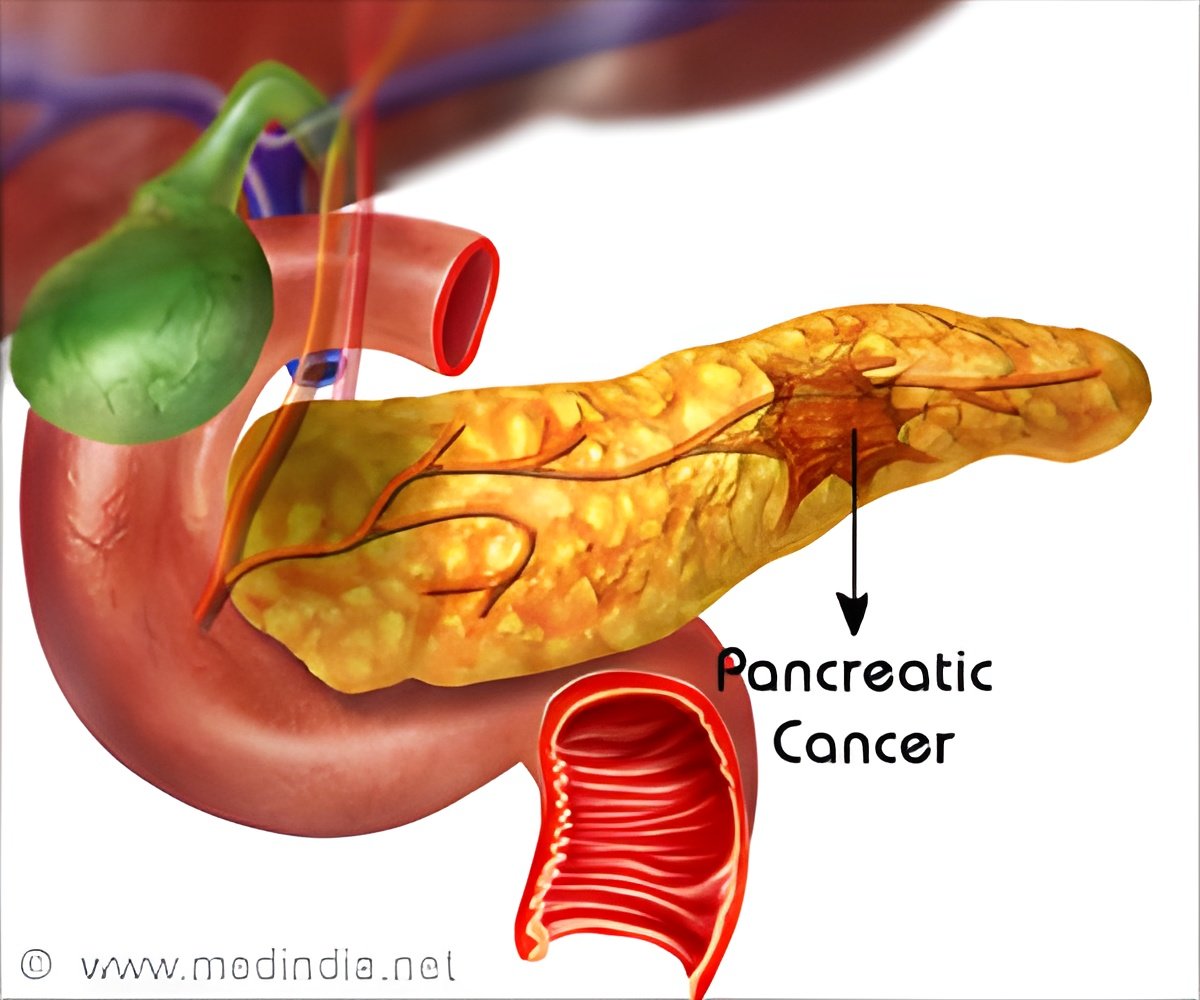
"Pancreatic cancer develops from these lesions, so if we understand how these lesions come about, we may be able to stop the cancer train altogether," says the study's lead investigator, Peter Storz, Ph.D., a cancer biologist.
The need for new treatment and prevention strategies is pressing, Dr. Storz says. Pancreatic cancer is one of the most aggressive human cancers -- symptoms do not occur until the cancer is well advanced. One-year survival after diagnosis is only 20 percent. It is the fourth leading cause of cancer death in this country.
The scientists studied pancreatic cells with Kras genetic mutations. Kras produces a protein that regulates cell division, and the gene is often mutated in many cancers. More than 95 percent of pancreatic cancer cases have a Kras mutation.
The researchers detailed the steps that led acinar cells with Kras mutations to transform into duct-like cells with stem cell-like properties. Stem cells, which can divide at will, are also often implicated in cancer.
They found that Kras proteins in the acinar cells induce the expression of a molecule, ICAM-1, which attracts macrophages, a specific kind of immune cells. These inflammatory macrophages release a variety of proteins, including some that loosen the structure of the cells, allowing acinar cells to morph into different types of cells. These steps produced the precancerous pancreatic lesions.
But the process can be halted in laboratory mice, he adds. "We could do this two ways -- by depleting the macrophages or by treating the transformed cells with a blocking antibody that shuts down ICAM-1," says Dr. Storz. "Doing either one reduced the number of precancerous lesions."
"Understanding the crosstalk between acinar cells with Kras mutations and the microenvironment of those cells is key to developing targeted strategies to prevent and treat this cancer," he says.
Source-Eurekalert
 MEDINDIA
MEDINDIA
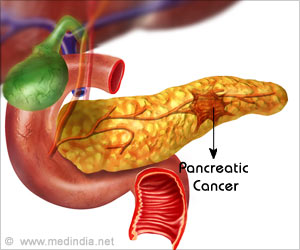


 Email
Email