Surgeons at the Ohio State University Medical Center have performed the first incisionless surgery in the US for gastroesophageal reflux disease (GERD) using a device known EsophyX.
Surgeons at the Ohio State University Medical Center have performed the first incisionless surgery in the US for gastroesophageal reflux disease (GERD) using a device known EsophyX. The Food and Drug Administration have approved the device for correcting GERD also known as heartburn.
The surgeons have treated the first two patients in the country by using the device.The mechanism, which is new in the US, has been proven safe and effective in patients treated throughout Europe for GERD.
According to the surgeons, the new procedure does not leave any scars and results in minimal post-operative pain. It also reduces patients’ recovery time significantly, they say.
“The medical device offers a treatment for patients who suffer from an advanced degree of GERD and, until now, would have been candidates for surgery,” said Dr. Scott Melvin, division director, general and gastrointestinal surgery, and director, Center for Minimally Invasive Surgery at the OSU Medical Center.
Heartburn, known to be linked with an increased risk of cancer of the oesophagus, is a condition in which acid refluxes from the stomach into the oesophagus, and causes burning sensation in the chest.
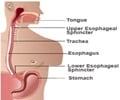
The incisionless surgery allows reconstruction of the one-way valve at the top of the stomach, when the valve is defective. During the procedure, the new tubular device is introduced to the body totally through the mouth, and is then advanced down the oesophagus into the stomach.
“Throughout medical history, we have gone from a very painful surgery, to a better tolerated surgery and, now, to a potentially pain-free incisionless procedure. In addition, the new procedure allows a treatment option for many patients when reflux is not severe enough to require surgery,” said Melvin.
Source-ANI
GAN/V
 MEDINDIA
MEDINDIA
 Email
Email