The US Food and Drug Administration on Tuesday warned pharmaceutical laboratories to stop marketing two unapproved products that have been linked to serious health problems.
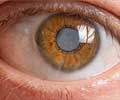
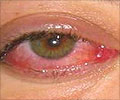
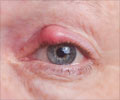
The first is an eye solution used to irrigate the eyes during and following cataract surgery, and the second is an ointment to treat sores and ulcers containing papain, which is not FDA approved."These unapproved products have put consumers health in jeopardy, from reports of permanent vision loss with unapproved balanced salt solutions to a serious drop in blood pressure and increased heart rate from the topical papain products," said Janet Woodcock, the FDA's director for the Center for Drug Evaluation and Research.
"Consumers need to be assured that all drug products are manufactured according to the high quality standards required for FDA approval and that they are safe and effective."
The FDA asked doctors to prescribe only ophthalmic balanced salt solutions (BSS) which are FDA-approved and produced by Alcon or Akorn.
Out of the some 3.2 million bottles of BSS solution sold in the United States since 2007, just one percent were approved by the FDA.
The FDA recommended that doctors refrain from prescribing any topical drug ointments containing the enzyme papain.
Advertisement
Source-AFP
SRM