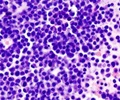
An experimental immunotherapy agent was put on the fast track to market approval by US regulators on Monday, after 89 percent of leukemia patients in early trials saw their cancers disappear.
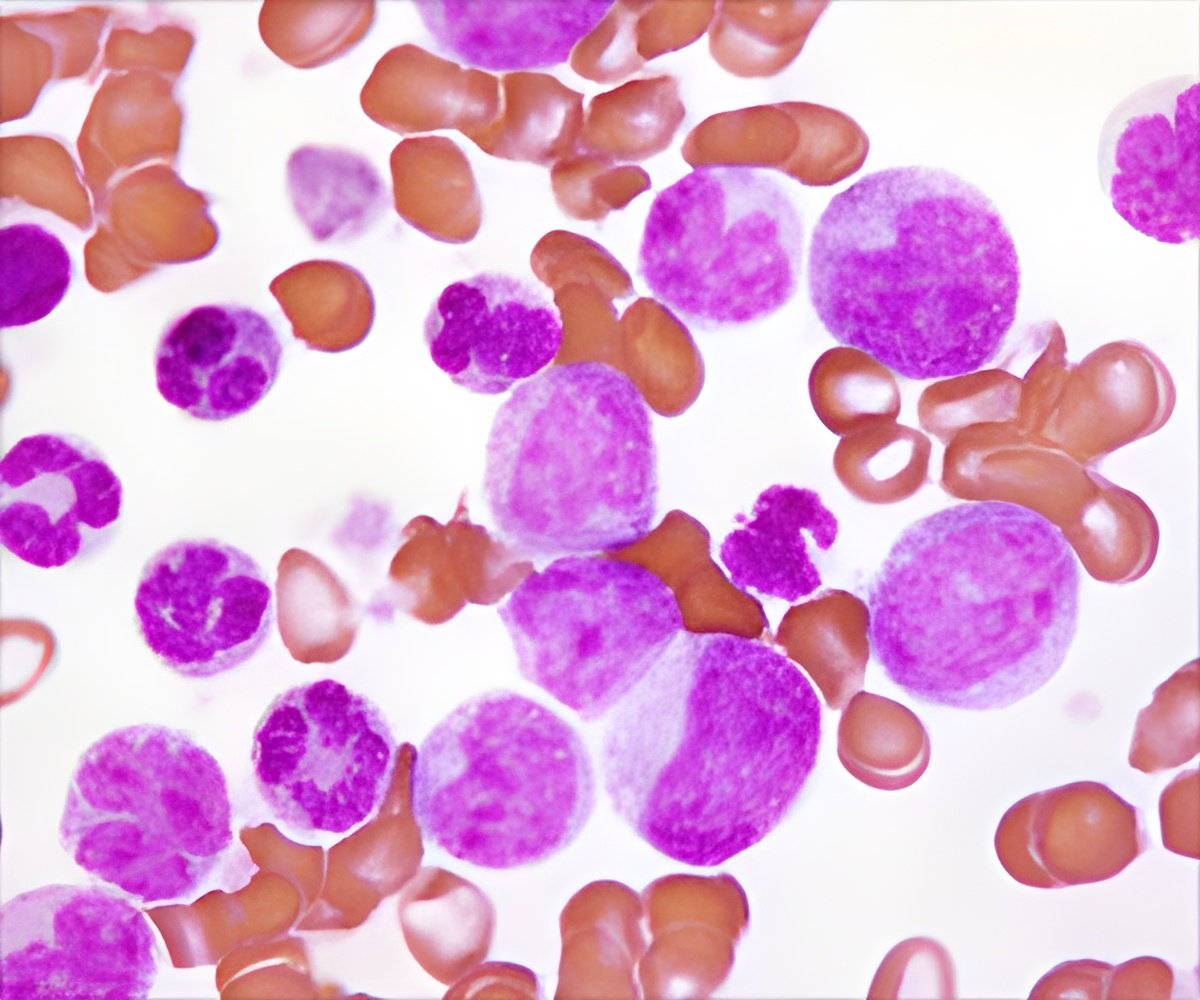
That means the experimental therapy will benefit from a speedier than average review process and will get extra attention from the FDA toward development for market.
It is the first cancer immunotherapy to receive the breakthrough designation, and only the fifth biologic agent so far.
The approach works by extracting a patient's T-cells, then genetically programming them in the lab to target cancer cells that produce a protein called CD19.
The altered T-cells are then re-injected into the patient's body, where they multiply and attack cancer.
Researchers reported last year that of 27 patients, including 22 children and five adults, with acute lymphoblastic leukemia, 89 percent had a complete response to the therapy, meaning their cancer became undetectable.
"Our early findings reveal tremendous promise for a desperate group of patients, many of whom have been able to return to their normal lives at school and work after receiving this new, personalized immunotherapy," said the Penn research team's leader, Carl June.
In addition to the ongoing trials for acute lymphoblastic leukemia, trials using CTL019 began in the summer of 2010 in patients with relapsed and refractory chronic lymphocytic leukemia.
It is also being tried in patients with non-Hodgkin lymphoma and myeloma.
More research is also under way into CAR therapies for mesothelioma, ovarian, breast and pancreatic cancers.
Source-AFP
 MEDINDIA
MEDINDIA


 Email
Email