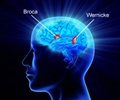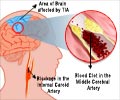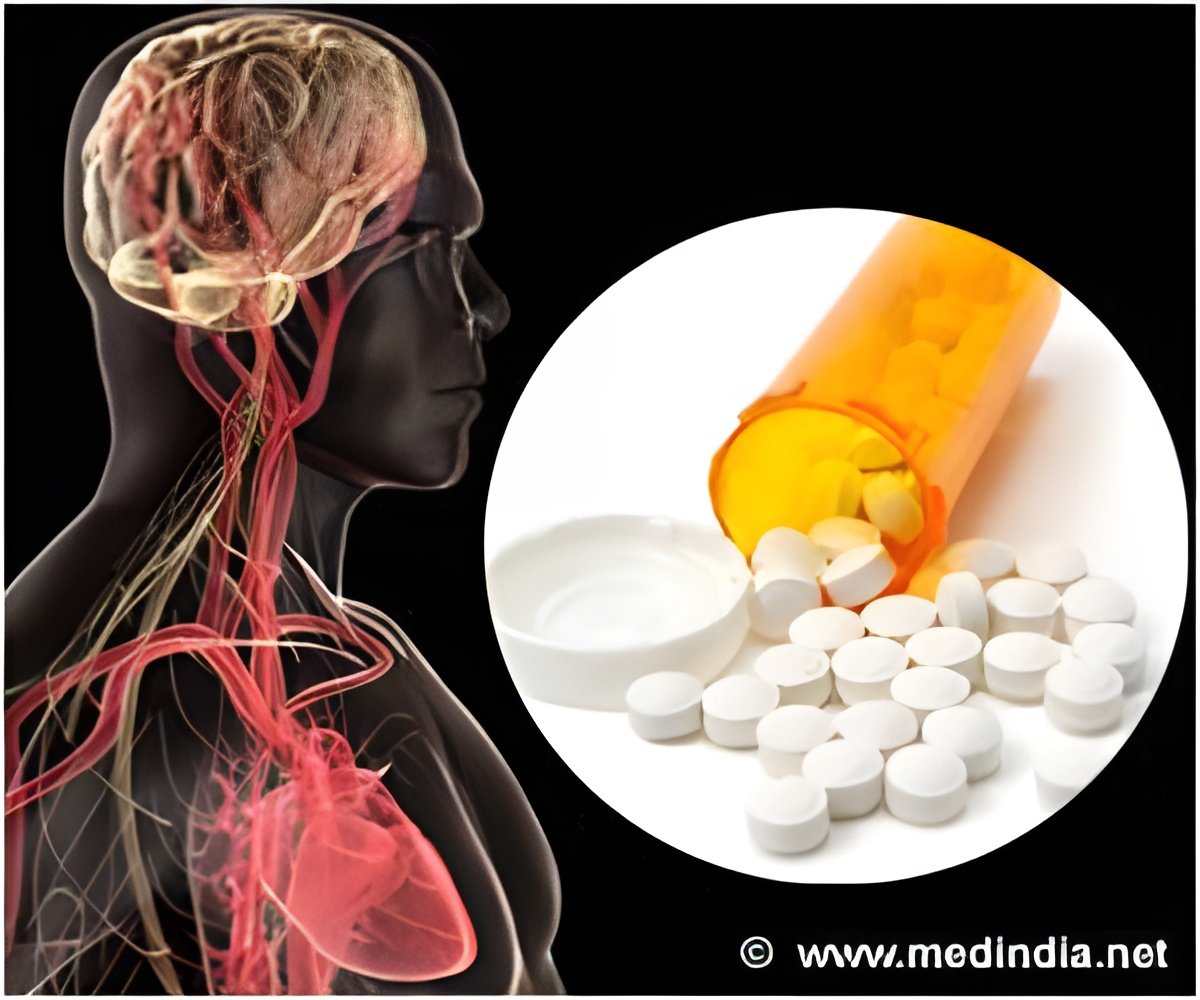
Stroke, which occurs when blood flow to a part of the brain stops, is the No. 4 cause of death and the leading cause of adult disability in the United States. According to the American Stroke Association, the Food and Drug Administration-approved tPA (tissue plasminogen activator) is the best treatment for stroke caused by a blocked artery, but to be effective, it must be administered within three hours after symptoms start. If given outside that three-hour window, tPA has shown serious side effects in animal and human brains, including bleeding and breakdown of the brain's protective barrier.
Generally, according to the American Stroke Association, only 3 to 5 percent of those who suffer a stroke reach the hospital in time to be considered for tPA treatment.
"What tPA does best is to break down clots in the blood vessel and restore blood flow, but it is a powerful enzyme," said Berislav V. Zlokovic, M.D., Ph.D., director of the Zilkha Neurogenetic Institute at the Keck School of Medicine of USC and the study's lead investigator. "After three hours, tPA also damages the blood vessel and causes intracerebral bleeding. We have developed something that not only counteracts the bleeding but also reduces brain damage and significantly improves behavior after stroke. I feel very strongly that this approach will extend the therapeutic window for tPA."
Zlokovic is the scientific founder of ZZ Biotech, a Houston-based biotechnology company he co-founded with USC benefactor Selim Zilkha to develop biological treatments for stroke and other neurological ailments. The company's 3K3A-APC is a genetically engineered variant of the naturally occurring activated protein C (APC), which plays a role in the regulation of blood clotting and inflammation. APC has cell-protecting, anti-inflammatory and anti-coagulant properties; 3K3A-APC has reduced anti-coagulant ability, which minimizes the risk of bleeding induced by normal APC. The protective effect of 3K3A-APC on the lining of blood vessels in the brain further helps prevent bleeding caused by tPA.
In collaboration with the University of Rochester Medical Center, Henry Ford Health Sciences Center, University of Arizona College of Medicine and The Scripps Research Institute, Zlokovic and his team gave tPA — alone and in combination with 3K3A-APC — to mice and rats four hours after stroke. They also gave 3K3A-APC for three consecutive days after stroke. They measured the amount of brain damage, bleeding and motor ability of the rodents up to seven days afterward.
Advertisement
"Dr. Zlokovic's study really demonstrates the promise of the drug and we are eager to show the same results in human clinical trials," said Kent Pryor, Ph.D., M.B.A., ZZ Biotech's chief operating officer.
Advertisement
"We are encouraged by these results," said Joe Romano, CEO and president of ZZ Biotech. "In terms of improving treatment for stroke and other neurological diseases, this could be really exciting."
Source-Eurekalert