Researchers have long known that the devastating disease called Duchenne muscular dystrophy (DMD) is caused by a single mutation in a gene called dystrophin.
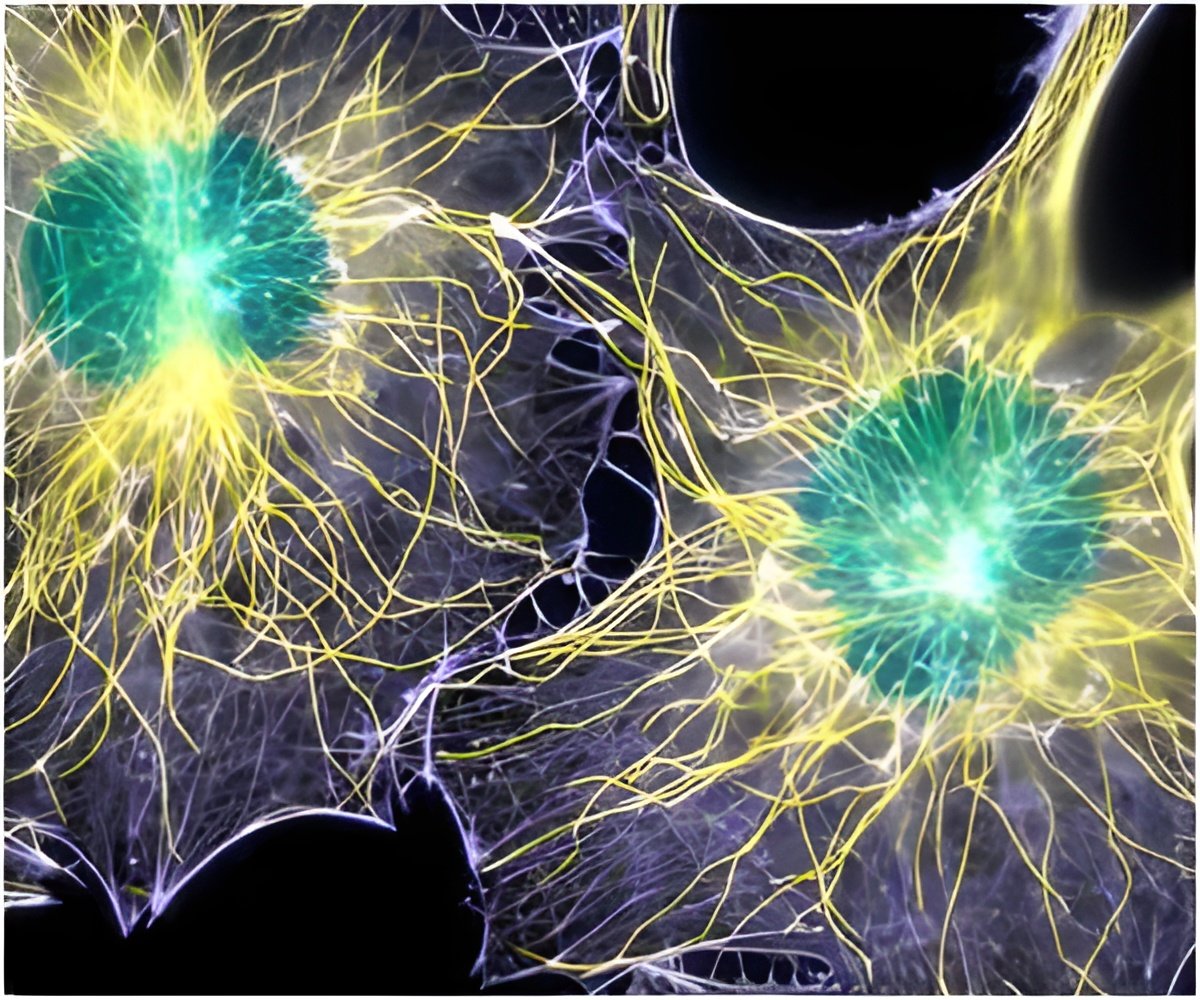
"These findings are critical for thinking about how to treat the disease and when," added Jason Pomerantz, the study's co-corresponding author who is now at the University of California, San Francisco. "It predicts any treatment designed solely to build muscle or enhance muscle function without replenishing the stem cell compartment is likely to fail and may even accelerate the decline. It's like pushing the gas pedal to the floor when there is no reserve."
The new study also answers a long-standing puzzle in the field that has stymied basic studies in search of potential treatments or treatment strategies: Mice carrying the same dystrophin mutation found in human patients show only mild symptoms of the disease.
"It has been a mystery for the past 25 years that mice with the genetic defect show minimal or no symptoms," Blau said, "and consequently there has been no mouse model in which to study the pathophysiology of the disease or potential treatments." People thought maybe it was because mice are smaller or don't live as long, but there was no real explanation. That is, until now.
The new findings attribute the discrepancy between the mouse and human symptoms to a characteristic of chromosomes. Regions of repetitive DNA found at the tips of chromosomes, known as telomeres, are longer in mice than they are in humans. Blau and her team have found that mice with the dystrophin mutation and another that leads them to have shortened telomeres have severe symptoms of the disease that worsen with age just as they do in human patients.
Telomeres protect chromosomes from deterioration and they tend to get shorter each time a cell divides. When telomeres become critically shortened, it triggers events that lead cells to die. The longer telomeres normally found in mice apparently give their muscle stem cells greater staying power and a greater capacity to repair the damage caused by the deficiency of dystrophin.
Advertisement
"There is continuous damage due to the loss of dystrophin," Pomerantz explained. "When the stem cell reserve is depleted, the symptoms emerge. The mice are spinning their wheels in a cycle of damage, repair, damage, repair, until the ability to repair gives out. In these mice [with shortened telomeres], it gives out earlier."
Advertisement
The mice are now the first tractable model system for studying the disease, and that should come as good news to families affected by this form of muscular dystrophy, the researchers say.
"Our new mouse model changed the way we were thinking about the pathophysiology of the disease," said Foteini Mourkioti of Stanford who is the co-first author on the paper. "We now understand that muscle stem cells are an essential component of this dystrophin-deficient disease and we can now start thinking of more precise ways to treat Duchenne muscular dystrophy."
Treatments intended to restore muscle will likely work only temporarily or not at all. In fact, they are likely to exacerbate the problem by exhausting muscle stem cells more rapidly. Timing will also be key.
"Therapeutic strategies aimed at intervening early in DMD patients, in the first years of their life, are more likely to have a better outcome as they would act before this end-stage tissue failure is reached," said Alessandra Sacco, the study's first author who is now at the Sanford-Burnham Medical Research Institute.
Source-Eurekalert