An Austrian government report charges that clinical trials conducted by researchers at a major medical school in Austria were riddled with procedural and ethical problems, the Nature
Clinical trials conducted by researchers at a major medical school in Austria were riddled with procedural and ethical problems, a new report by the Austrian government has charged, the Nature Publishing Group has revealed.
The Agency for Health and Food Safety report says University of Innsbruck Professor Hannes Strasser failed to get necessary approvals for the trials, and neglected to adequately inform patients about the nature of the procedure, according to Nature.Strasser, who designed the study, is also charged with failing to insure the volunteers.
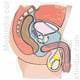
The trials tested a new treatment for urinary incontinence on hundreds of women and men. The therapy involved removing tissue from a patient's arm to create muscle stem cells, then injecting these cells into the same person's urinary sphincter muscle.
Findings were published last year in the British medical journal The Lancet.
A lawyer representing some of the patients said many had experienced serious side effects, such as the sealing of the urinary sphincter and bladder rupture, according to Nature, a leading scientific journal based in Britain.
"This is a very grave scandal for the university, and the pressure on the public prosecutor to bring criminal charges will be very high," the lawyer, Thomas Juen, is quoted as saying.
The university hospital has barred Strasser -- who has written an open letter to university authorities denying any wrongdoing -- from treating patients.
Georg Bartsch, head of the university's urology department, was cleared of wrongdoing in the report even though he is listed in the study published in The Lancet as one of five co-authors who did "all investigations and treatments" related to the therapies, Nature said.
The Lancet said in a statement it was reviewing the government report, and was in touch with both Austrian authorities and the authors of the report.
Source-AFP
RAS/S
 MEDINDIA
MEDINDIA


 Email
Email





