Tumors in melanoma patients create conditions that knock out the body's 'premier' immune defence, new research reveals.
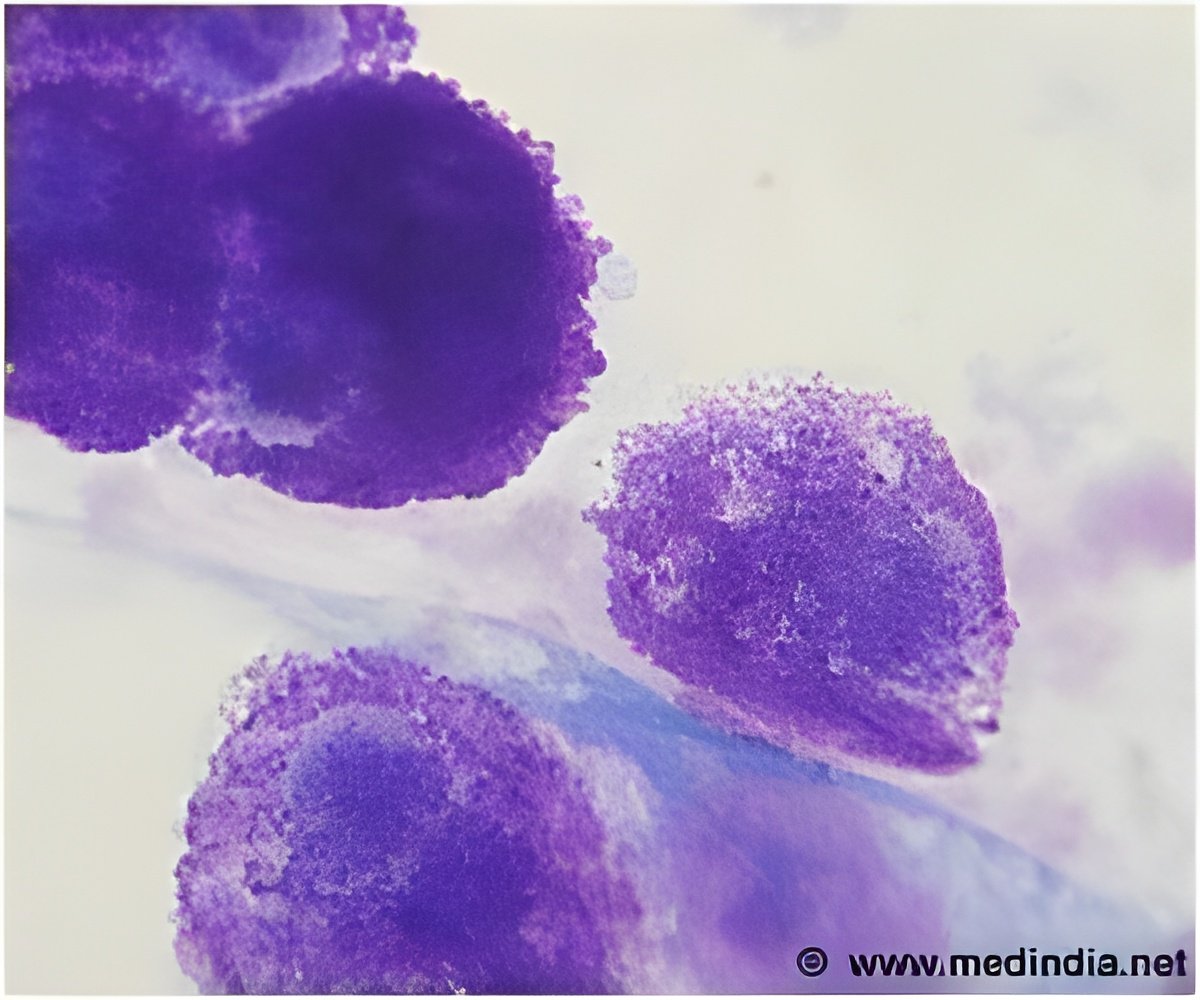
Karagiannis and colleagues have previously shown that, in patients with melanoma, antibodies are produced that can attack tumour cells. Despite this, the patient's immune system is often ineffective in preventing the cancer from progressing.The body's B cells (part of the immune system) produce a total of 5 different antibody classes. The most common, IgG, comprises 4 types (or subclasses) of which the researchers have shown that IgG1 subclass antibodies are the most effective at activating immune cells, while antibodies of the IgG4 subclass are thought to be the least efficient. In this new research, the authors analysed tumour tissue and blood donated by 80 patients from the melanoma clinic of St John's Institute of Dermatology at Guy's and St Thomas', as well as tissue and blood from healthy volunteers.By analysing the lesions found in melanoma, the authors show that melanoma tumours not only create conditions that attract IgG4, the weakest possible response, but also that IgG4 antibodies interfere with the action of any IgG1 antibodies circulating. "We were able to mimic the conditions created by melanoma tumours and showed that B cells can be polarised to produce IgG4 antibodies in the presence of cancer cells," says Dr Karagiannis. In the presence of healthy cells, the body's immune response functions normally, and IgG1 are the main antibodies circulating. To better understand the functional implications of IgG4 subclass antibodies in cancer, the authors engineered these two antibodies (IgG1, IgG4) against a tumour antigen and demonstrated that unlike IgG1, the IgG4 antibody was ineffective in triggering immune cells to kill cancer cells. Importantly, IgG4 also blocked the tumour cell killing actions of IgG1, thus preventing this antibody from activating immune cells to destroy tumours. Additionally, using samples from 33 patients, the authors found that patients with higher IgG4 levels in their blood are more likely to have a less favourable prognosis compared to those whose blood levels of IgG4 are closer to normal levels. This suggests that IgG4 may help assist in predicting disease progression."This work bears important implications for future therapies since not only are IgG4 antibodies ineffective in activating immune cells to kill tumours but they also work by blocking antibodies from killing tumour cells," says Dr Karagiannis. "The latter means that IgG4 not only prevents the patient's more powerful antibodies from eradicating cancer, but could also explain why treatments may be hindered by those native IgG4 antibodies found in patients, making therapeutic antibodies less effective.""Now, with the help of our NIHR Biomedical Research Centre, more work needs to be done on developing IgG4 as a potential clinical and prognostic biomarker which can improve patient care by informing clinical decisions and helping to identify patients most likely to respond to treatments," concludes Professor Nestle. Therefore, these findings are expected to inform the design and help improve the potency and efficacy of future therapies for cancer. "This study can also inform the rational design of novel strategies to counteract IgG4 actions."The authors are now broadening the study by examining larger groups of patients. The team is analysing blood and sera from patients with melanoma and from patients with other cancers to determine whether the presence of IgG4 could inform patient outcomes or predict responses to therapy. They are also analysing the mechanisms of IgG4 blockade of new and existing therapeutic antibody candidates, and developing new antibody candidates which may be less prone to IgG4 blockade.
Source-Eurekalert
 MEDINDIA
MEDINDIA

 Email
Email





