Effects of anti-CTLA-4 antibody on the number and types of T cells present in patient's blood was investigated using LymphoSIGHT™ immune repertoire sequencing platform.
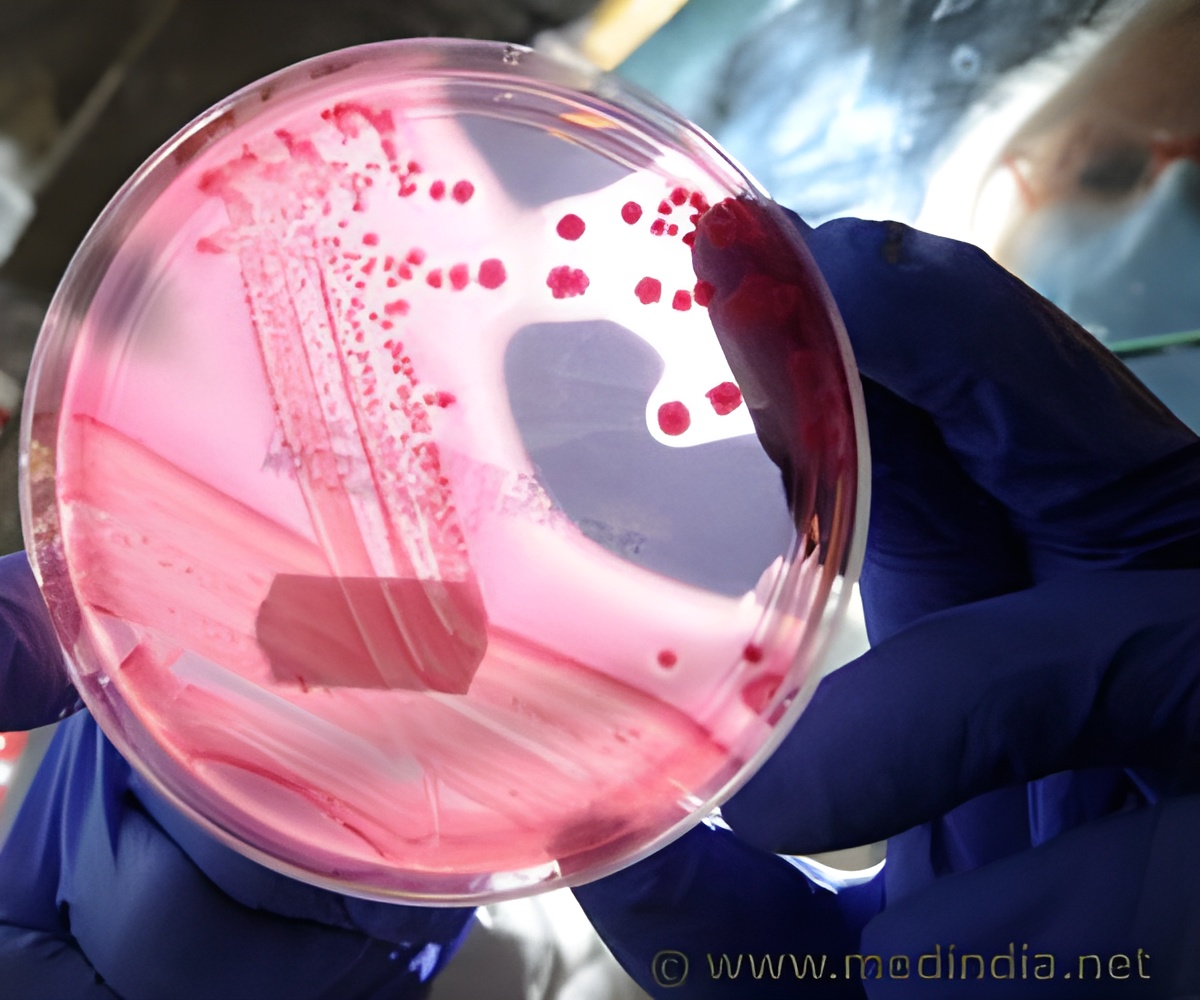
The researchers used next-generation sequencing to assess the impact of CTLA-4 blockade on changes to the T cell repertoire in 25 metastatic castration-resistant prostate cancer patients treated with ipilimumab and GM-CSF, 21 metastatic melanoma patients treated with tremelimumab, and nine untreated control subjects. Results showed that patients with longer survival had significantly lower numbers of T cell clones (sets of T cells that all express the same receptor) that decreased in frequency after treatment compared to patients with shorter survival. This association held for both the prostate cancer patients (p < 0.01) and the melanoma patients (p < 0.05).
"By leveraging the power of Sequenta's LymphoSIGHT platform for immune repertoire profiling, we and our collaborators have uncovered new aspects of the mechanism of action of anti-CTLA-4 monoclonal antibodies in human cancers," said Tom Willis, CEO of Sequenta. "We have also demonstrated the potential of next-generation sequencing analysis of the immune repertoire for identification of patterns that may predict clinical benefit of immunotherapies, which will be crucial for optimal use of currently available treatments and efficient development of new ones."
Source-Eurekalert
 MEDINDIA
MEDINDIA


 Email
Email










