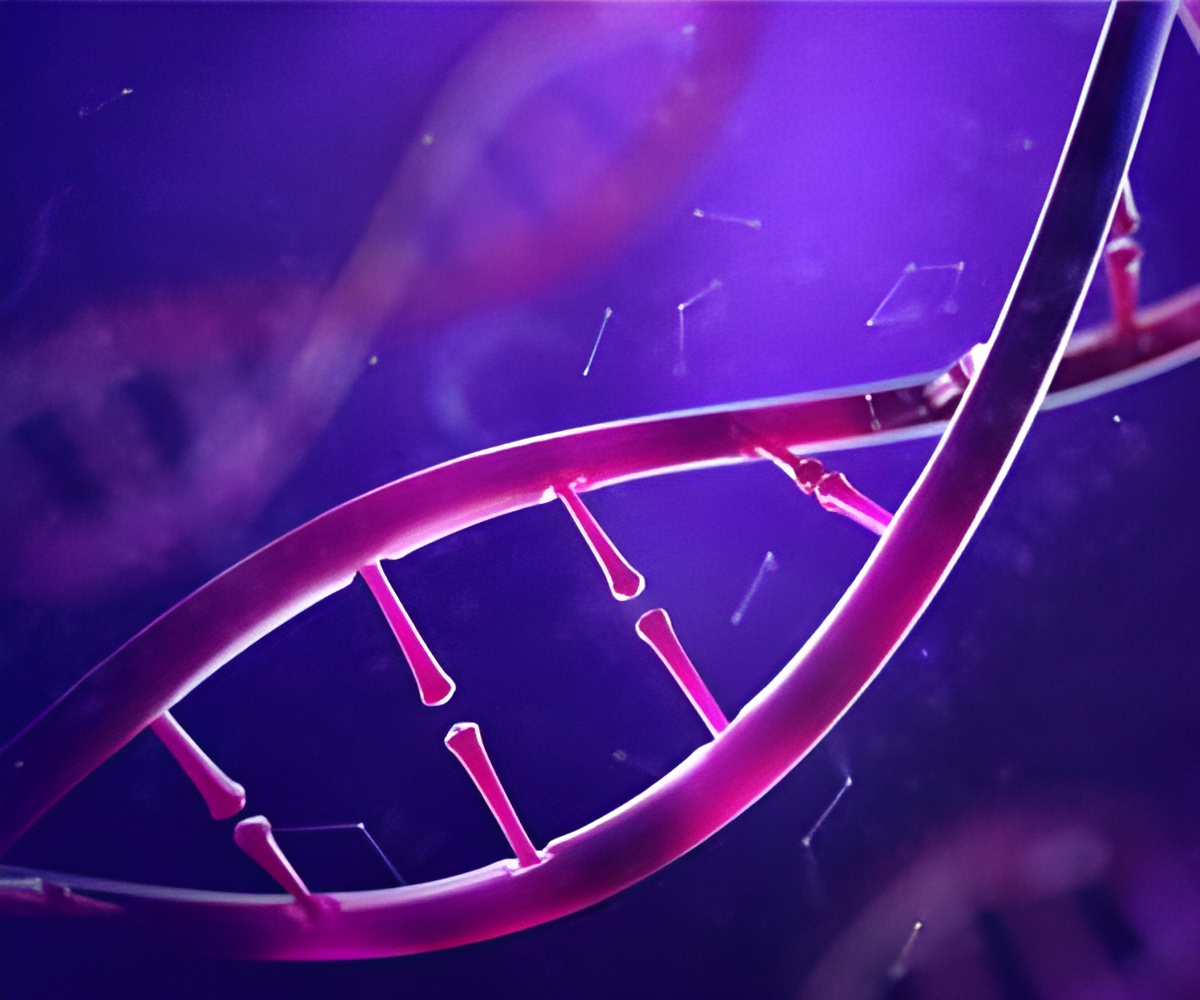
The ability to detect and analyze cancer DNA from a patient's blood sample is emerging as a promising alternative to invasive surgical biopsies.
TOP INSIGHT
ichorCNA can analyze DNA fragments for mutation patterns and detect cancer.
"And, as tumors evolve in more advanced stages of cancer, developing resistance or becoming metastatic, we might access timepoints that could be pivotal in deciding which therapies are right for that patient."
This ability to detect and analyze cancer DNA from a patient's blood sample is emerging as a promising alternative to invasive surgical biopsies, which can be difficult, painful, and costly -- especially when tumors have appeared in locations that are challenging to access.
Blood biopsies (also called liquid biopsies) are poised to overcome many of these issues. They have the potential to allow doctors to track the progress of disease and treatment in real-time and to help researchers understand how tumors resist treatment with far greater resolution.
Understanding cancer without invasive procedures
Tracking this data could make it possible to monitor cancer recurrence, a patient's response to treatment, and other clinically important features, all from blood samples.
Compiling a whole exome from DNA fragments currently requires at least 10 percent tumor DNA in a blood sample, but the fraction of tumor DNA in the blood can vary wildly from patient to patient. Because of this variation, the team first desired an unbiased approach for detecting and measuring levels of cancer DNA before attempting whole-exome sequencing.
Across the field, many blood biopsy methods detect tumor DNA by screening for mutations in known cancer-related genes, but this targeted sequencing misses cancers without those mutations.
Co-first author Ha, a postdoctoral scholar at the Broad Institute and Dana-Farber Cancer Institute (DFCI), led the development of a tool called ichorCNA that can analyze DNA fragments for mutation patterns nearly universal in cancer genomes, and as a result capture cancers with both known and unknown mutations. Ha focused on detecting stretches of DNA that have either fewer or greater copies in cancer cells, in contrast to healthy cells.
The research team tested and refined ichorCNA on 1,439 blood samples collected prospectively from 520 metastatic breast or prostate cancer patients at DFCI (a significant effort championed by medical oncologists Atish Choudhury, Daniel Stover, Heather Parsons, Nikhil Wagle, and colleagues).
Using this approach, the researchers found that in 33 to 49 percent of the metastatic breast and prostate cancer patients, depending upon whether one or multiple blood samples were examined, tumor DNA made up greater than 10 percent of the cell-free DNA in their blood -- enough to make whole-exome sequencing of cell-free DNA feasible.
Then, to determine whether this sequencing of cell-free tumor DNA could offer the same level of insight into cancer genetics as a tissue biopsy could, the team compared surgically obtained tumor biopsies to data collected from whole-exome sequencing of cell-free DNA from a group of 41 patients.
The researchers found that genetic data from blood whole-exome sequencing and tissue biopsies matched significantly across a number of genetic features, such as clonal somatic mutations (88 percent match) and copy number alterations (80 percent match).
These results support cell-free DNA whole-exome sequencing, from blood samples, as a potential substitute for metastatic tumor biopsy sequencing for many patients.
"Our study has demonstrated that we can get a cancer whole exome reliably, from blood; that it reflects the matched tumor biopsy; and that it can be done for a significant fraction of patients with metastatic cancer," said Adalsteinsson.
"This validation suggests that we can use blood biopsies for large-scale genomic characterization of disease in patients with metastatic cancer."
"It unlocks the potential for a lot of studies that we couldn't do before," added Getz, institute member and director of the Cancer Genome Computational Analysis group at Broad and associate professor of Pathology and director of Bioinformatics at the Massachusetts General Hospital Cancer Center and Department of Pathology.
"The technology will allow us to track the dynamics of cancer and understand the evolution of drug resistance, or the development of the metastatic state, in a way that isn't possible through surgical biopsies."
A means to perform large-scale blood biopsies could allow researchers and clinicians easy access to the cancer genome, with exciting implications for the way physicians monitor response to treatment, watch for recurrence, and more. The ability to frequently and non-invasively monitor cancer and its treatment could alter clinical trials, increase the resolution with which clinicians understand metastatic cancer, and potentially increase accessibility to quality precision medicine approaches.
"Using cell-free DNA to track cancer is not a new idea, but we're developing the tools to understand how we can better qualify materials for those types of analyses, and we're doing it in a way that allows us to look across the genome broadly," said Love, a Broad associate member, associate professor of chemical engineering at MIT, and a member of the Koch Institute for Integrative Cancer Research at MIT.
"We've established quality metrics to make sure that this technology is cost-effective and scalable for thousands of patients and samples a year."
Source-Eurekalert
 MEDINDIA
MEDINDIA


 Email
Email










