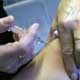
British drugmaker GlaxoSmithKline will next week begin shipping its swine flu vaccine Pandemrix across Europe, the group announced on Wednesday following approval by regulators.
"Following the (European) Commission's approval we expect to commence shipments of Pandemrix to European governments from next week," said Jean Stephenne, President of GlaxoSmithKline Biologicals, in a company statement.The news came one day after the European Commission cleared Pandemrix and another vaccine, Focetria, for distribution in Europe. Focetria is produced by Swiss pharaceuticals group Novartis.
"GSK confirmed today that the European Commission has granted marketing authorisation of GSK's pandemic (H1N1) adjuvanted vaccine Pandemrix for protection against pandemic H1N1 2009 influenza," the group added Wednesday.
The decision on the two vaccines was pushed through last week by the European Medicines Agency amid deep concern about the new winter influenza season and an upturn in British cases of swine flu.
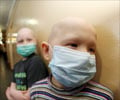
Health experts have meanwhile warned of a second wave of flu in the coming months as the northern hemisphere winter approaches.
The commission had announced Tuesday that the vaccines will be authorised for use in all 27 EU member nations as well as Iceland, Liechtenstein and Norway. The authorisation was expected to take effect by October 1.
Advertisement
In Britain -- the hardest hit European country by the virus -- authorities aim to begin vaccinations in October.
Advertisement
Source-AFP
LIN