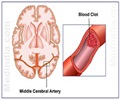
A new study has revealed that an old intravenous antibiotic may act as a potential new treatment for stroke.
Lead researcher Dr. David Hess, chair of the Department of Neurology in the Medical College of Georgia School of Medicine said that an old drug known as minocycline may reduce stroke damage in multiple ways by inhibiting white blood cells and enzymes that, at least acutely, can destroy brain tissue and blood vessels.The antibiotic may also reduce cell suicide in the minutes and hours following a stroke, enabling more cells to recover.
"It's a safe drug that is easy to give and tolerate, that gets into the brain well, and may reduce bleeding, the primary side effect of tPA. We think it will make strokes smaller and patient outcomes better," said Dr Hess.
The tPA drug is the only FDA-approved drug therapy for strokes. The team conducting a clinical trial that will study the drug in 60 stroke patients in Georgia, Kentucky and Oregon say they believe the antibiotic will be a safe, effective adjunct therapy for tPA, the only FDA-approved drug therapy for strokes.
The studies conducted on animals have shown the drug, given within six hours of a stroke, then every 12 hours for up to three days, the peak time of inflammation, can reduce stroke damage by up to 40 percent.
"We know it's safe in humans and we know the concentrations we need to see improvement in the brains of rats can be achieved safely in humans," said Dr. Susan C. Fagan, professor of pharmacy at the University of Georgia, assistant dean for the MCG program of the UGA College of Pharmacy and study co-investigator.
Advertisement
The participants will be receiving one of four doses, starting with 200 milligrams, the most common dose already used, and increasing incrementally up to 700 milligrams and half their first dose at subsequent 12-hour intervals for a three-day period then be followed for 90 days.
Advertisement
They would be looking in the blood for biomarkers, indicators of inflammation, to see if inflammatory factors go up after three days, which could provide a clue on how to treat patients.
Dr Hess said that Minocycline fights inflammation is by inhibiting microglial cells, white blood cells activated by a stroke and also blocks apoptosis, or cell suicide.
"We wanted something we could give at least three hours after stroke or later," said Fagan.
Source-ANI
SRM