Mechanism that causes chronic graft-versus-host disease after bone marrow transplant has been identified. Expression of miR-17-92 in donor T- and B-cells is essential for the induction of both scleroderma and bronchiolitis obliterans in cGVHD.
TOP INSIGHT
One particular family of microRNAs (miRs), called miR-17-92, is responsible for the T-cell and B-cell pathogenicity that causes chronic graft-versus-host disease.
GVHD can be divided into acute (aGVDH) or chronic forms (cGVHD). "They are very different diseases," explains Yongxia Wu, Ph.D., a postdoctoral fellow and lead author on the article. "Our ability to prevent or treat aGVHD has considerably improved, but the incidence of cGVHD continues to increase. Chronic GVHD has a different pathophysiology and different target organs than aGVHD. It's been a big challenge to try to find a target for cGVHD therapies, because of the more complex immune reaction in cGVHD and the fact that its cellular and molecular mechanisms are not as well understood."
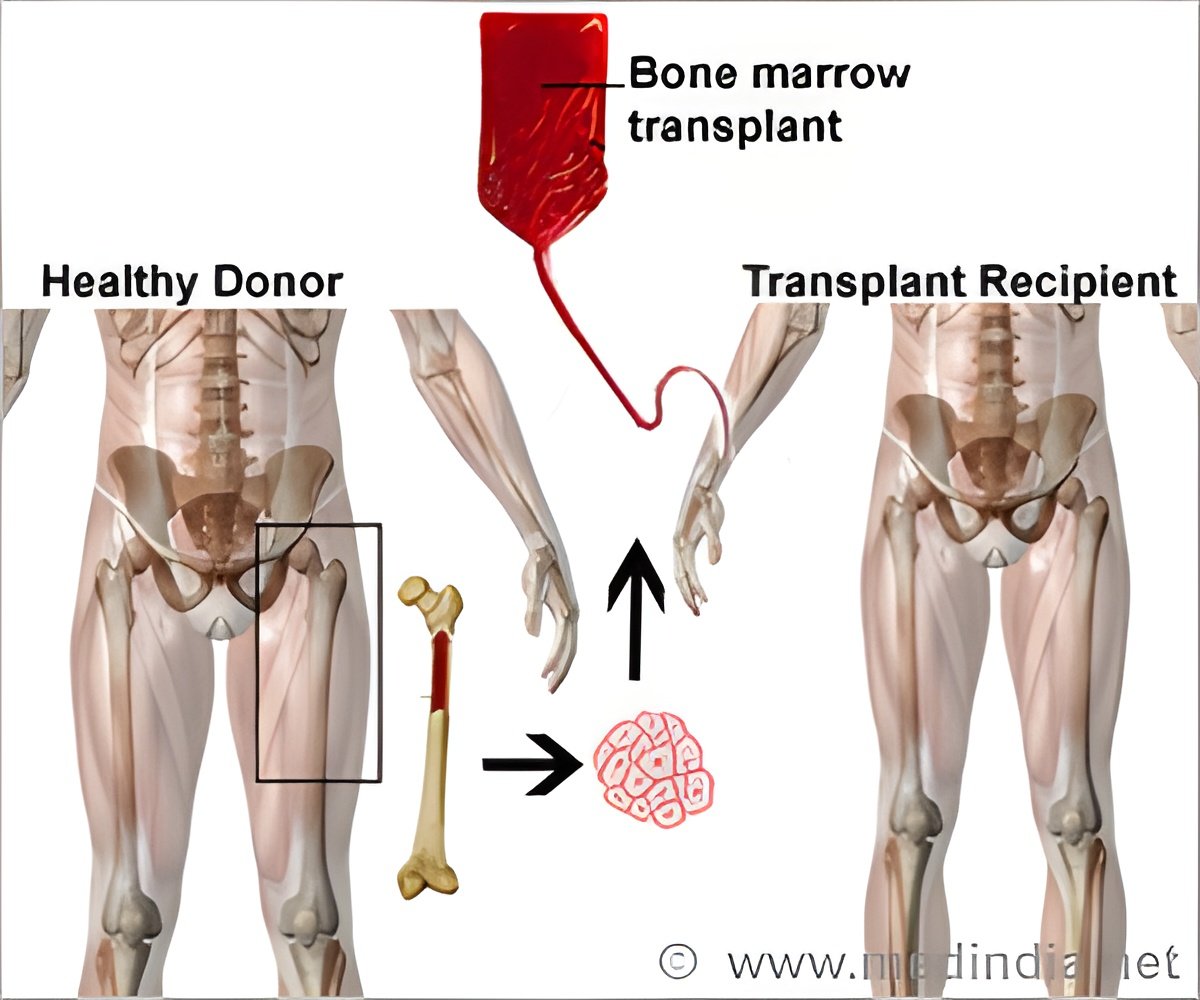
Chronic GVHD is characterized by autoimmune-like, fibrotic changes in multiple organs such as the skin (causing scleroderma) and the lungs (causing bronchiolitis obliterans), and fibrosis of the salivary glands, liver, and gut. With 30 to 70 percent of patients who receive allogeneic BMT developing cGVHD, the lack of effective therapies is a major unmet clinical need.
The MUSC team previously found that, in acute GVHD, miR-17-92 played a critical role in regulating CD4 T-cell proliferation and Th1 and Treg differentiation. Based on this work, they decided to investigate whether miR-17-92 regulates T- and B-cell differentiation and function in the development of cGVHD.
"We decided to extend our aGVHD study to cGVHD. But there's no single, well-defined murine model that can reflect all of the clinical manifestations seen in cGVHD patients," explains Wu. "Different patients experience different symptoms because cGVHD can be manifested in many organs -- some patients have skin symptoms, some have lung symptoms -- it varies. So, we decided to study four different cGVHD models to best understand how miR-17-92 contributes overall, across many clinical presentations."
Their results demonstrated shared mechanisms by which miR-17-92 mediates cGVHD progression -- namely by regulating T helper-cell differentiation, B-cell activation, germinal center responses, and autoantibody production. The clinical translation studies also found that miR-17 blockade alleviated proteinuria (in the lupus-like condition) and scleroderma symptoms.
Although miR-17-92 has been well studied, its role in cGVHD development has never before been defined. Because cGVHD has a similar pathophysiology to some autoimmune diseases, it is likely that these findings will be useful for developing new treatments and preventive therapies in other conditions.
"We are very excited to publish this work because we are hoping that a clinical research group will be inspired to take our study findings further in patients," says Wu.
In the meantime, the MUSC team, led by Yu, will continue their work and try to extend the current findings by investigating how other miRs may be involved in regulating T- and B-cell function during allogeneic BMT.
Source-Eurekalert
 MEDINDIA
MEDINDIA
 Email
Email




