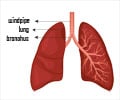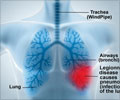
Stating that a large section of the population over 50 years of age are suffering from diseases caused by pneumococcus bacteria, the FDA revealed that the latest approval will provide an alternative option for physicians to prevent such diseases.
“Pneumococcal disease is a substantial cause of illness and death. Today's approval provides an additional vaccine for preventing pneumococcal pneumonia and invasive disease in this age group”, the director of FDA's Center for Biologics Evaluation and Research, Dr Karen Midthun said.
Source-Medindia