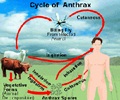
Panacea Biotech has bagged the registration, production and marketing rights of the anti-anthrax vaccine developed by the Centre for Biotechnology in the Jawaharlal Nehru University (JNU). It is expected that the vaccine would be in the market in 3-4 months. The Company has made a milestone payment to JNU for the exclusive rights. In addition, it will pay the university a royalty on sales. However, the company is yet to make any projections on the business the vaccine is expected to generate.
The vaccine is different from drugs such as ciprofloxacin currently being used in the treatment of anthrax. Ciprofloxacin is administered after a person has contacted anthrax. The vaccine develops immunity in the human body to anthrax bacillus. According to marketing director Rajesh, the vaccine developed by JNU is superior to the only other anti-anthrax vaccine available in the US. The vaccine in the US has a lot of toxic side- effects. The side-effect profile of this (JNU) vaccine is negligible. As anthrax has become a national emergency, the government's policy of fast-track registration will come into effect. This, in turn, will lead to a speedy commercialisation of the vaccine.Panacea Biotech To Market Anthrax Vaccine
Recommended Readings
Advertisement