"We carried out a highly sophisticated analysis of MYC activity in cells, but came away with a simple rule. MYC is not a power switch but a universal amplifier," said co-lead study author Keji Zhao, Ph.D., director of the Systems Biology Center at the NIH's National Heart, Lung, and Blood Institute (NHLBI). "This discovery offers a unifying idea of how and why abnormal levels of MYC are found in so many different cancer types, such as breast cancer, lung cancer, and several blood cancers."
"MYC is much like the volume control of a music player," added co-lead David Levens, M.D., Ph.D., a senior investigator in the Laboratory of Pathology at the National Cancer Institute (NCI), also part of NIH. "If you're listening to opera, for example, adding more MYC will make the opera louder, but it won't change the program to rap. And if you have only silence, MYC will just give you more silence."
Both researchers noted that this new understanding of MYC function could influence future treatment efforts for MYC-associated tumors. They suggest that trying to limit MYC activity, or turning down the volume just the right amount, would be a better strategy than using targeted chemotherapy to try to eliminate all MYC activity.
MYC aids in cell activation, a process in which cells mature and divide quickly. During an immune response, for example, white blood cells are activated to help fight infections. If activation isn't properly regulated, then cells can start growing out of control and result in cancer. Researchers have known that abnormally high levels of MYC can lead to cancer, but until now, no one had been able to explain how it can lead to so many different cancers.
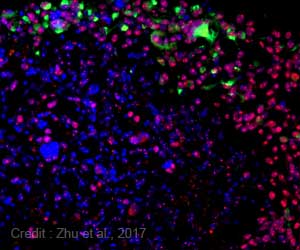
Zhao, Levens, and their colleagues used a specially designed fluorescent protein that allowed them to track MYC in white blood cells in a lab dish. They chose white blood cells, specifically B cells and T cells that fight infections, because they are frequently affected by abnormal MYC and can transform into lymphoma and myeloma cells.
Advertisement
The analysis revealed that MYC didn't prefer any specific type of gene. Instead, MYC proteins were present at nearly every gene that was already expressed, or turned on. The researchers also noticed that the amount of MYC at each expressed gene correlated with how active that gene was prior to immune stimulation. The more active the gene, the more MYC gathered there. MYC appeared to amplify productivity relative to the initial expression levels where it gave a small boost to genes with low activity and a big boost to genes with high activity.
Advertisement
The research team then conducted the same analysis in embryonic stem cells and got similar results.
Source-Eurekalert