‘FDA-approved immunosuppressive medication anakinra could protect the brain cells from inflammation and reverses the neurodevelopmental abnormalities caused by Zika virus.’
Read More..Tweet it Now
However, in the Zika-infected mouse experiments, the drug appears to interfere with inflammation in the pregnant animals' placentas, the researchers say. There also is evidence the drug directly reduces inflammation in fetal brains. Read More..
A report on the findings was published in the April issue of the Journal of Clinical Investigation Insights.
"Until now, the focus of research has been on finding vaccines and antiviral drugs, but our study strongly suggests that the placental immune response should not be overlooked as a target for treatment," says Irina Burd, M.D., Ph.D., associate professor of gynecology and obstetrics and director of the Integrated Research Center for Fetal Medicine at the Johns Hopkins University School of Medicine. "Using an FDA-approved drug already shown to be safe in infants shortens the time that we may be able to quickly start clinical trials and get a potentially effective preventative measure approved and available to help decrease the harmful effects of Zika."
According to the U.S. Centers for Disease Control and Prevention, 10% of babies born in the U.S. to women with a Zika infection during pregnancy develop fetal brain birth defects that range from slow head growth to microcephaly, a condition marked by a very small head due to brain abnormalities.
Zika can be spread by infected mosquitos or unprotected sex from an infected person, and is passed from a pregnant woman to her unborn child. Pregnant women with Zika are also at increased risk for miscarriage.
Advertisement
For this study, the researchers first wanted to see how Zika may affect the placenta in pregnant mice with the virus to try to figure out the cause behind the fetal deaths and birth defects. They compared pregnant mice infected with strains of Zika found in Nigeria, Puerto Rico and Brazil to pregnant mice not infected with the virus.
Advertisement
Because Zika causes early overproduction of interleukin-1beta, the researchers turned to the drug anakinra to test its potential for alleviating the damaging effects to fetal mice of mothers with Zika.
The researchers injected mice placentas with either 10 milligrams per kilogram of anakinra or with fluid without the drug.
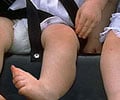
After eight days of in utero exposure to the virus, 1.8% of the 322 mice infected with Zika but not given the drug had birth defects such as contracted limbs with tightened muscles, kinked tails, and fused fingers and toes. None of the mice given the drug showed signs of these birth defects.
Comparable to the defects shown in the mice, human babies with microcephaly-caused defects may have limb contractures. The researchers say it is important to note that the defects noted in the mice may vary from humans because each species develops differently.
The researchers also say that more fetal mice treated in utero with anakinra survived to full-term birth compared to untreated mice. In the pregnant mice with Zika, 39.2% of the mothers had fetal deaths. In mice with Zika given the drug in utero, 20.8% of the mothers had fetal deaths.
Next, the researchers wanted see if the drug protected the brains of the mice born to mothers with Zika from inflammation. The researchers took mice immune system cells from the brain, called microglia, and infected them with Zika. After a day, microglia with Zika had made more cells, showing that the Zika was causing inflammation.
They treated microglia infected with Zika with anakinra, and after 24 hours there were fewer microglia, suggesting that the drug protected the brain cells from inflammation.
Five days after birth, the mice born treated with anakinra completed neurological and physical assessments that tested balance, movement, vision, depth perception and coordination. For example, in the coordination test, the mice were put on their backs and researchers measured how long it would take them to flip over.
Mice given the drug flipped over to become upright about one second faster on average than mice without treatment, which the researchers say was significant for this kind of test. The researchers believe the drug was responsible for reversing the neurodevelopmental abnormalities that caused longer performance times.
"Currently, there is no cure for Zika, but our study suggests that there may be FDA-approved medications like anakinra that have the potential to combat some of the worst effects of the virus," says Sabra Klein, Ph.D., associate professor of molecular microbiology and immunology at the Johns Hopkins University Bloomberg School of Public Health. "
"Future studies are urgently needed to determine the possible benefits of such a drug in people," says Burd. "More and more countries are affected by Zika. And it would be great for us to continue doing this research with the same rigor and funding so we can continue to fight this not only in the United States but, globally."
Source-Eurekalert