- Thalassemias - (http://www.nhlbi.nih.gov/health/health-topics/topics/thalassemia/)
- Deferiprone - (http://www.nlm.nih.gov/medlineplus/druginfo/meds/a612016.html#side-effects)
- Bone Marrow for Hemoglobinopathy Research - (http://www.clinicaltrials.gov/show/nct00669305\)
- What is the difference between thalassemia minor and major? - (http://www.medicinenet.com/beta_thalassemia/article.htm#what_is_the_difference_between_thalassemia_minor_and_major)
- Beta Thalassemia - (http://emedicine.medscape.com/article/206490-overview)
- BMT Thalassemia - (http://www.curethalassemia.org/bone-marrow-transplantation-bmt/)
- U.S. Gene Therapy Trial for Thalassemia Receives FDA Approval - (http://www.thalassemia.org/u-s-gene-therapy-trial-for-thalassemia-receives-fda-approval-2/)
- What is thalassemia? - (http://www.healthline.com/health/thalassemia#symptoms3)
About
Thalassemia is an inherited blood disorder passed on through parental genes causing the body to produce abnormal hemoglobin.
Thalassemia, also known as Cooley’s anemia, was first described by the paediatrician Thomas Benton Cooley in 1925. thalassemia cases are distributed in the Mediterranean region, India, parts of South-Asia, parts of South-East Asia and the Middle East, Europe, America and Australia. The term thalassemia was first coined by George Whipple, the Nobel Prize winning pathologist. Thalassa in Greek means the sea and –emia means of the blood so thalassemia refers to anemia around the sea.
Hemoglobin A, which is the normal haemoglobin, has four protein chains: two alpha globin and two beta globin. Four genes (two from each parent) are required to make sufficient alpha globin protein chains and alpha thalassemia (minor/major) occurs when one/two of the four chains are missing. Two genes (one from each parent) are required to make sufficient beta globin protein chains and beta thalassemia (minor/major) occurs when one or both genes are mutated.
Minor and Major Thalassemia
- Minor Thalassemia: is the heterozygous state wherein one of the parents is a carrier of the mutated gene. The individual has only one copy of the thalassemia gene along with one normal beta-globin chain gene. Individuals with thalassemia minor do not require any treatment except nutritional supplements and lead a fully normal life.
- Major Thalassemia: is the homozygous state wherein both parents are carriers of the mutated gene. The individual has two copies of the thalassemia gene. Individuals with thalassemia have severe anemia requiring lifelong blood transfusions and other medical procedures for survival.
Causes and risk factors associated with thalassemia:
- One/both parents carrying the mutated beta-globin chain gene.
- Endogamous/cross-cousin marriages.
What are the symptoms thalassemia?
Symptoms of thalassemia are usually seen before the child is two years of age. Anemia occurs in the first few months after birth and becomes progressively severe. Early diagnosis and treatment can prevent fatality.
The most common signs and symptoms of thalassemia include paleness, fussiness, jaundiced complexion, susceptibility to infections, low appetite and enlarged organs (usually the spleen).
At birth, a baby with thalassemia may seem normal because the haemoglobin at this stage is mostly fetal haemoglobin (HbF). Since HbF has only two alpha chains and two gamma chains (unlike haemoglobin A which has two alpha and two beta chains) the baby does not show any symptoms of thalassemia. However, anemia starts occurring in the first few months after birth. The baby may become pale and tired, often failing to feed properly from exhaustion, and has frequent episodes of fever, vomiting and diarrhea.
Symptoms of thalassemia include:
- Paleness
- Fussiness
- Jaundice (yellowed complexion due to breakdown of abnormal hemoglobin)

- Frequent infections
- Low appetite
- Enlarged organs (usually the spleen)
How is thalassemia diagnosed?
The first diagnosis for thalassemia is usually via a Complete Blood Count (CBC) and hemoglobin electrophoresis.
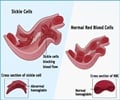
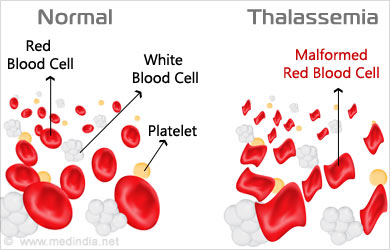
Doctors usually diagnose thalassemia with a Complete Blood Count (CBC) and hemoglobin electrophoresis. Pathologists may examine the Red Blood Cells (RBCs) to check if the RBCs are misshapen. Abnormal RBCs are a sign of thalassemia. The hemoglobin electrophoresis separates out the different molecules in the RBCs aiding in identifying the abnormal type.
Tests used in the diagnosis of thalassemia :
- Blood tests: Complete Blood Count (CBC) and hemoglobin electrophoresis.

- Genetic analysis: to check for the beta-globin chain gene mutation.
What is the Available Treatment for thalassemia?
Blood transfusion, iron-chelation and a non-iron diet along with dietary supplements comprise available treatment.
Prior to commencement of any line of treatment, a thorough DNA testing is required to decide the accurate line of treatment because each individual varies in DNA patterns.
Blood transfusion of packed cells is the line of treatment available to treat anemia in thalassemia. Blood is cross-matched with the individual’s blood group and washed, packed, leucocyte-poor cells are preferred for blood transfusion. The blood should be screened stringently for HIV-1, HIV-2, HTLV-1, HTLV-2, Hepatitis-B, Hepatitis-C, and syphilis along with compatibility testing (COOMBS test). Children and adults with thalassemia usually require two units of blood every two weeks.
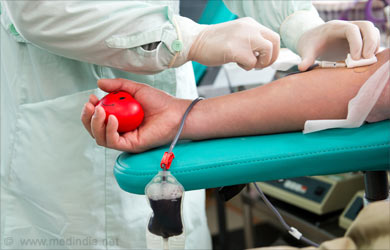
Apart from regular blood transfusion, individuals with thalassemia require regular iron-chelation therapy because repeated and frequent blood transfusions lead to iron build-up in the blood and vital organs. Iron overload can cause serious damage to the cardiac, hepatic, musculo-skeletal and endocrine systems leading to complications like arrhythmia, impaired liver function, bone malformations, arthritis, diabetes and impaired reproductive system.
Iron-chelation therapies available are:
- Deferoxamine: Is a subcutaneous infusion via pump given nocturnally over 8-10 hours per night. The number of vials required is calculated on the individual’s body weight and serum ferritin levels.
- Deferasirox: Is an oral chelator in the form of a daily pill taken once-a-day with water or fruit juice. Some of the side effects of the oral chelator are usually vomiting, diarrhea and occasional joint pains.
- Deferiprone: Is another iron chelator available as tablets/capsules to be taken usually three times a day. Some of the side effects include White Blood Cell (WBC) reduction, nausea, vomiting, diarrhea, heartburn, stomach pain and joint pain.
Apart from regular blood transfusion and iron-chelation, it is advised to be on a low-iron diet avoiding foods rich in non-soluble iron like red meat, jaggery, and ragi (finger millet). Greens like spinach are allowed as the iron is water-soluble. Drinking weak tea during mealtimes helps prevent iron absorption. Vitamin C is recommended as also folic acid depending on the individual’s age and body weight.
In many cases, doctors also recommend a spleenectomy to remove the spleen to prevent the individual from losing RBCs. Spleenectomy also implies a lower immune system as the spleen usually fights certain types of bacteria. In these cases, doctors recommend regular vaccines like pneumococcal vaccine to prevent pneumonia, influenza vaccine, hepatitis A and B vaccines and typhoid vaccine.
Long-term treatment options for thalassemia include:
- Bone marrow transplant: from a compatible donor (usually a matched sibling or matched unrelated donor. Risks include susceptibility to infections and rejection of host marrow.
Future Therapies
At present, the US FDA (Food and Drug Administration) has approved gene therapy trial in which blood stem cells will be extracted from the individual’s system and then introduced with a vector containing a functional beta-globin gene. The vector here is a disabled viral vector. Low-dose chemotherapy will be given to the individual to suppress his/her immune system and finally the genetically engineered stem cells will be infused back. The clinical trials are still on at Memorial Sloan Kettering, Boston and St. Jude’s hospital, Memphis. This therapy is indeed the most promising curative for thalassemia.