Causes
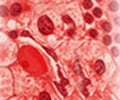
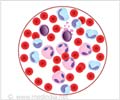
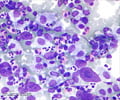
95% of CML patients report the presence of a specific marker, the Philadelphia (Ph) chromosome.
CML is characterized by the presence of a specific marker, a chromosomal abnormality called the Philadelphia (Ph) chromosome. Approximately 95% of those suffering from CML harbor this abnormality. The Ph chromosome results from a translocation of
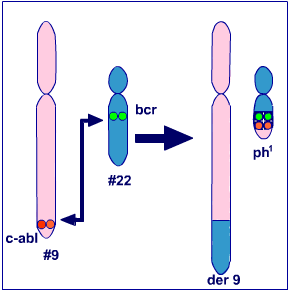
[Schematic representation of 9:22 translocation. der9 -> chromosome 9 with extra translocated segment; ph1 -> Philadelphia chromosome.]
The 9:22 translocation brings two different genes together- the breakpoint cluster region gene (BCR),on chromosome 22 and the Ableson leukemia virus gene (ABL), a proto- oncogene, on chromosome 9. The resulting hybrid gene, the BCR-ABL codes for a protein, endowed with tyrosine kinase activity, which has the ability to activate signal transduction pathways. This protein fosters uncontrolled cell proliferation and genomic instability.
In mouse models a CML-like disease has been induced on administering bone marrow cells infected with a BCR-ABL gene-containing virus. In other animal models, the fusion proteins have been shown to transform normal progenitor blood cells to malignant cells.
The other risk factors involved in the onset of CML, irrespective of the Philadelphia chromosome, are the following:
- Exposure to high amounts of ionizing radiation is the most prominent risk factor for CML. For instance, increased rates of CML were observed in people who were exposed to the atomic bombings of Nagasaki and Hiroshima.
- CML is more common among men than in women
- CML mostly affects older people between the ages of 50 to 60 years.