- Collins MH, Metz SW. Progress and works in progress: update on flavivirus vaccine Development. Clin Ther. 2017 Jul 25. pii: S0149-2918(17)30768-3. doi - (10.1016/j.clinthera.2017.07.001. PMID: 28754189.)
- García LL, Padilla L, Castaño JC. Inhibitors compounds of the flavivirus replication process. Virol J. 2017 May 15;14(1):95. doi - (10.1186/s12985-017-0761-1. PMID: 28506240.)
- Basu A, Dutta K. Recent advances in Japanese encephalitis. F1000Res. 2017 Mar 13;6:259. doi - (10.12688/f1000research.9561.1. PMID: 28357054.)
- Vrati S, Bharati K. Chapter 110: Japanese encephalitis. In: Talwar GP, Hasnain SE, Sarin SK, editors. Textbook of Biochemistry, Biotechnology, Allied and Molecular Medicine. 4th ed. New Delhi: PHI Learning Private Limited; 2016. p. 1280-90. - (10.12688/f1000research.9561.1. PMID: 28357054.)
- Leibovitch EC, Jacobson S. Vaccinations for neuroinfectious disease: a global health priority. Neurotherapeutics. 2016 Jul;13(3):562-70. doi - (10.1007/s13311-016-0453-3. PMID: 27365085.)
- Chokephaibulkit K, Houillon G, Feroldi E, Bouckenooghe A. Safety and immunogenicity of a live attenuated Japanese encephalitis chimeric virus vaccine (IMOJEV®) in children. Expert Rev Vaccines. 2016;15(2):153-66. doi - (10.1586/14760584.2016.1123097. PMID: 26588242.)
- World Health Organization. Japanese encephalitis. Fact sheet No. 386. December 2015. - (10.1586/14760584.2016.1123097. PMID: 26588242.)
- Pavli A, Maltezou HC. Travel-acquired Japanese encephalitis and vaccination considerations. J Infect Dev Ctries. 2015 Sep 27;9(9):917-24. doi - (10.3855/jidc.5108. PMID: 26409731.)
- Lord JS, Gurley ES, Pulliam JR. Rethinking Japanese encephalitis virus transmission: a framework for implicating host and vector species. PLoS Negl Trop Dis. 2015 Dec 10;9(12):e0004074. doi - (10.1371/journal.pntd.0004074. PMID: 26657648.)
- Erra EO, Kantele A. The Vero cell-derived, inactivated, SA14-14-2 strain-based vaccine (Ixiaro) for prevention of Japanese encephalitis. Expert Rev Vaccines. 2015;14(9):1167-79. doi - (10.1586/14760584.2015.1061939. PMID: 26162529.)
- Ishikawa T, Konishi E. Potential chemotherapeutic targets for Japanese encephalitis: current status of antiviral drug development and future challenges. Expert Opin Ther Targets. 2015;19(10):1379-95. doi - (10.1517/14728222.2015.1065817. PMID: 26156208.)
- Wang H, Liang G. Epidemiology of Japanese encephalitis: past, present, and future prospects. Ther Clin Risk Manag. 2015 Mar 19;11:435-48. doi - (10.2147/TCRM.S51168. PMID: 25848290.)
- Chen HL, Chang JK, Tang RB. Current recommendations for the Japanese encephalitis vaccine. J Chin Med Assoc. 2015 May;78(5):271-5. doi - (10.1016/j.jcma.2014.12.009. PMID: 25841620.)
- Firbas C, Jilma B. Product review on the JE vaccine IXIARO. Hum Vaccin Immunother. 2015;11(2):411-20. doi - (10.4161/21645515.2014.983412. PMID: 25621812.)
- Monath TP, Seligman SJ, Robertson JS, Guy B, Hayes EB, Condit RC, Excler JL, Mac LM, Carbery B, Chen RT; Brighton Collaboration Viral Vector Vaccines Safety Working Group (V3SWG). Live virus vaccines based on a yellow fever vaccine backbone: standardized template with key considerations for a risk/benefit assessment. Vaccine. 2015 Jan 1;33(1):62-72. doi - (10.1016/j.vaccine.2014.10.004. PMID: 25446819.)
- Yun SI, Lee YM. Japanese encephalitis: the virus and vaccines. Hum Vaccin Immunother. 2014;10(2):263-79. doi - (10.4161/hv.26902. PMID: 24161909.)
- Li X, Ma SJ, Liu X, Jiang LN, Zhou JH, Xiong YQ, Ding H, Chen Q. Immunogenicity and safety of currently available Japanese encephalitis vaccines: a systematic review. Hum Vaccin Immunother. 2014;10(12):3579-93. doi - (10.4161/21645515.2014.980197. PMID: 25668666.)
- Daep CA, Muñoz-Jordán JL, Eugenin EA. Flaviviruses, an expanding threat in public health: focus on dengue, West Nile, and Japanese encephalitis virus. J Neurovirol. 2014 Dec;20(6):539-60. doi - (10.1007/s13365-014-0285-z. PMID: 25287260.)
- Ankur Nandan V, Nilesh K, Dibyaranjan B, Ashutosh T, Ravi A, Arvind A. Acute transverse myelitis (ascending myelitis) as the initial manifestation of Japanese encephalitis: a rare presentation. Case Rep Infect Dis. 2013; 2013: 487659. doi - (10.1155/2013/487659. PMID: 23585973.)
- Ratnam I, Leder K, Black J, Biggs BA, Matchett E, Padiglione A, Woolley I, Panagiotidis T, Gherardin T, Luxemburger C, Torresi J. Low risk of Japanese encephalitis in short-term Australian travelers to Asia. J Travel Med. 2013; 20: 206-8. doi - (10.1111/jtm.12019. PMID: 23577868.)
What is Japanese Encephalitis?
Japanese encephalitis (JE) is so called because it was first reported from Japan and the major complication is encephalitis (inflammation of the brain). JE is the most important form of viral encephalitis, commonly known as “Brain Fever” that affects young children and adolescents and is endemic in most parts of Southeast Asia, China, India, and Oceania. JE is caused by a virus called Japanese encephalitis virus (JEV) and is transferred from infected animals - usually pigs and wading birds - to humans, through the bite of infected Culex mosquitoes. The incubation period of Japanese encephalitis is 5-15 days. Majority of infections are asymptomatic. Only 1 in 250 infections develop into encephalitis.
The onset of this disease is marked by severe rigors. It is soon followed by altered mental status, seizures, tremors, headache, neck stiffness and/or sensitivity to light.
There is no specific treatment for Japanese encephalitis. No specific antiviral drug is available. Treatment aims at relieving the symptoms like pain and reducing the intracranial pressure. Some patients may require long-term care and rehabilitation for residual neurologic deficits.
Japanese encephalitis can be prevented by vaccination. WHO recommends vaccination for JE to be part of the routine vaccines in national immunization schedules in all areas where the disease is recognized as a public health issue.
What are the Types of Japanese Encephalitis Infections?
JE infections can be of two types:
1. Primary Infection: Primary infection is caused by direct infection by the virus and is more serious.
2. Secondary Infection: Secondary encephalitis is due to a post-infectious immune reaction to viral infection elsewhere in the body and is more common. However, because of the milder nature of secondary encephalitis, more cases of primary encephalitis are seen.
Facts on Japanese Encephalitis
Japanese encephalitis virus (JEV) is a flavivirus and comes under the same class of viruses as those that cause dengue, yellow fever and West Nile viruses.
The flavivirus is usually prevalent in the rural areas of many countries in Asia (China, Sri Lanka, Philippines, Nepal, Indonesia and South East Asia) with epidemics breaking out every few years.
There is endemic JEV transmission in 24 countries of WHO South-East Asia and Western Pacific regions, exposing more than 3 billion people to risk of infection.
It is the leading cause of viral encephalitis in Asia with 68,000 clinical cases reported annually.
Fatality rate among those with encephalitis can be as high as 30%.
Permanent consequences of neurologic or psychiatric injuries can occur in 30%–50% of those with encephalitis.
What are the Causes of Japanese Encephalitis?
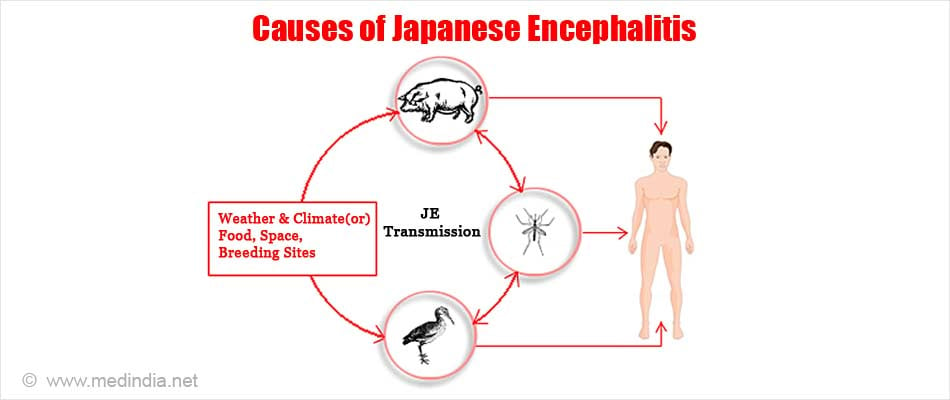
Japanese encephalitis is caused by the Japanese encephalitis virus (JEV), which belongs to the family Flaviviridae. Since the encephalitis virus is transmitted via arthropod vectors, it is an arbovirus and the disease is an arbovirus infection.
JEV is spread by mosquitoes, predominantly, Culex tritaeniorhynchus, which breeds particularly in flooded rice fields. Birds are often the source of infection for mosquitoes, which can then spread the infection to horses and other animals.
Man is an incidental host. Since man-to-man transmission of the disease is not possible, humans are also regarded as “dead-end hosts”.
What are the Symptoms of Japanese Encephalitis?
Signs and symptoms that require urgent care may include altered levels of consciousness. In infants, the key signs are a stiff neck and a bulging in the fontanels of the skull. In older children, severe headaches, lethargy, confusion, hemiparesis (weakness on one side of the body), opisthotonus (arching of the body as in tetanus), dystonia (abnormal muscle tone resulting is muscle spasm in the affected body part), and sensitivity to light can occur.
In adults, mental disturbances may be more prominent.
Most people infected with Japanese encephalitis have no symptoms or have only mild symptoms. These are listed below:
- Headache
- Irritability
- Lethargy
Serious cases of Japanese encephalitis can cause:
- Severe rigors
- Sudden fever
- Severe headache
- Drowsiness
- Confusion and disorientation
- Seizures
- Nausea and vomiting
- Tremor or convulsions
- Stiff neck (occasionally)
- Bulging in the fontanels of the skull in infant

How can we Diagnose Japanese Encephalitis?
The diagnosis is made primarily on the basis of the patient's symptoms, history of mosquito bites and the knowledge of the diseases endemic to a particular geographic region. Therefore, the doctor should keep in mind other viral diseases that have a similar clinical presentation to JE. There include herpes simplex virus (HSV) encephalitis and West Nile encephalitis.
Physical Examination:A thorough physical examination is crucial in order to assess the level of consciousness and presence of any seizures arising from infection, which should be treated immediately.
Investigation of the Cerebrospinal Fluid (CSF): If the condition of the patient permits, a lumbar puncture can be done to obtain CSF to measure various parameters. For example, the opening pressure, cell count, glucose and protein levels can point to an infection of the central nervous system (CNS).
Laboratory tests to pinpoint the type of infection (viral or bacterial) and identify the causative microbe are also available.
Rapid Diagnostic Tests (RDT): Confirmation of JE infection requires laboratory diagnosis such as virus isolation or detection of IgM in the patient’s serum or CSF. The IgM antibody develops early, usually within 7 days, of the infection. This can be detected by the IgM antibody capture-enzyme-linked immunosorbent assay (MAC-ELISA). Currently, three MAC-ELISA kits are available:
- JEV CheX Kit (XCyton Diagnostics Ltd.)
- JE IgM ELISA (InBios International Inc.)
- JE-Dengue IgM Combo ELISA (Panbio Ltd.)
These tests can give a result within a matter of hours.
Molecular Tests: These tests include reverse transcription-polymerase chain reaction (RT-PCR), which can detect the genetic material of the virus and is therefore very specific. However, this requires a good lab to carry out and a machine in which high reaction temperatures (as high as 100ºC) can be generated. Another technique called reverse transcription-loop-mediated isothermal amplification (RT-LAMP) assay can detect JE at the same temperature (much lower than in RT-PCR) and thus would be useful for low-resource settings. RT-LAMP is a very fast technique and can give results with 30 minutes.
Imaging Techniques: Computed tomography (CT scan) and magnetic resonance imaging (MRI) can be used to detect changes in certain regions of the brain due to JE infection, and to correlate them with the symptoms of the patient.
How is Japanese Encephalitis Treated?
There is no specific treatment for Japanese encephalitis. The treatment is targeted at symptomatic relief of the patients.
Supportive Care: Most patients require supportive care as no effective antiviral drug has been discovered. Patients often require feeding and ventilation support. Intravenous (IV) fluids may be given to maintain the hydration status of the patient. Painkillers can be given to reduce the associated pain. Anticonvulsants are given to control seizures. The patient’s brain’s electrical activity should be monitored from time to time by carrying out electro-encephalography (EEG).
Reduction of Intracranial Pressure: Raised intracranial pressure is one of the most life-threatening complications of JE infection. Mannitol is commonly used to decrease intracranial pressure. Sometimes diurectics like Furosemide, which increase urine outflow, are also given along with Mannitol to bring down the pressure.
Some patients may require long-term care and rehabilitation for residual neurologic deficits, including seizures and movement disorders.

Current Research in the Development of Antivirals: Currently a lot of research is going on to develop antivirals that inhibit various stages of the virus replication cycle as well as various enzymes that catalyze the replication process. Some of these are listed below:
- Polymerase inhibitors
- Protease inhibitors
- Virus entry inhibitors
- Capping inhibitors
- Helicase inhibitors
Japanese Encephalitis Vaccine
Currently, there are three JE vaccines available to people. These are briefly highlighted below:
- SA14-14-2: This is a live-attenuated JE vaccine, based on the SA14-14-2 strain of JEV and has been developed by the Chengdu Institute of Biological Products, China. This vaccine is licensed for use in China, India, Sri Lanka, Nepal, and South Korea. This is a single-dose vaccine and is very cheap and therefore suitable for use in economically weaker countries in Asia, where JE is endemic.
- IXIARO®: This is a killed or inactivated JE vaccine, based on the SA14-14-2 strain of JEV and has been developed by Intercell, Austria. This is a two-dose vaccine and is licensed in North America, Europe, Hong Kong, Singapore, Israel, Australia, and New Zealand. This vaccine is recommended for travelers visiting JE endemic countries.
- IMOJEV®: This is a live-attenuated, genetically-engineered JE vaccine. It was initially developed by Acambis, UK and currently manufactured by Sanofi Pasteur, France. This is a single-dose vaccine and is used in most countries around the globe.
- Side-Effects: The JE vaccines could cause some mild side-effects in susceptible individuals. These include pain or tenderness at the injection site, headache or muscle ache.