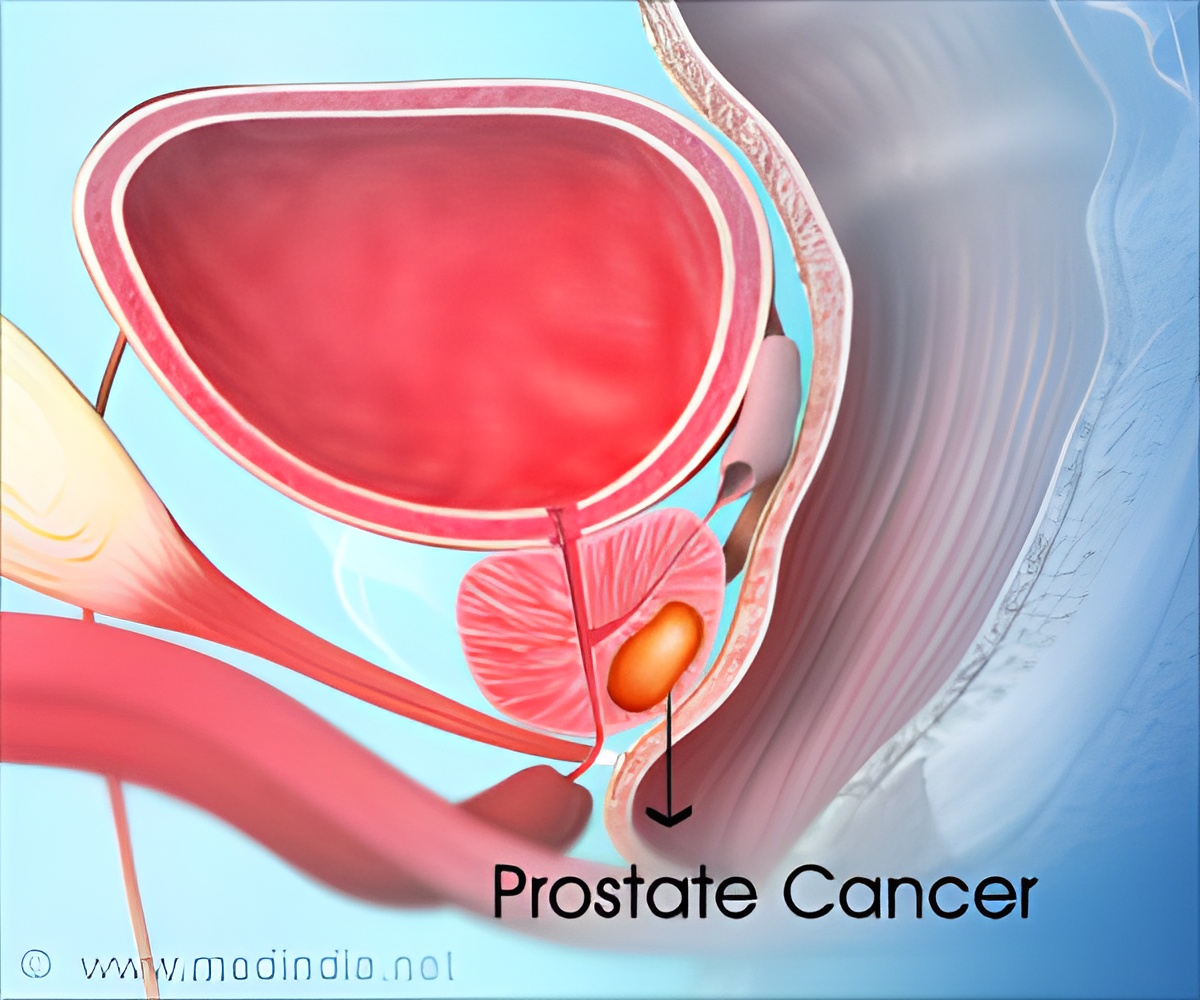
Professor Mary Gospodarowicz, President of the Union of International Cancer Control (UICC) welcomes the draft guidelines, saying: “The current PSA test is not accurate enough for population screening, yet it remains in widespread use. We need to focus on helping both patients and healthcare professionals understand the risks and benefits of this test to empower them to make more informed choices around screening. We applaud the Australian cancer authorities for taking the lead in developing the strongest set of clinical guidelines seen to date in prostate cancer; developed through a truly multisectoral approach. We expect the final recommendations of this review to clear the current confusions that exist around PSA testing.”
Approaches to prostate cancer testing differ across the world. Decision-making is complicated by the need to balance effective cancer detection and care, with concerns about over-diagnosis and prostate cancer treatment adverse side effects, including urinary incontinence, loss of sexual potency in men treated by radiotherapy and bowel problems.
The new draft guidelines, developed under the auspices of Cancer Council Australia’s guidelines unit with funding from Prostate Cancer Foundation of Australia (PCFA), address these risk/benefit concerns. Key recommendations include:
For men without a prostate cancer diagnosis or symptoms that might indicate prostate cancer
- For men informed of the benefits and harms of screening who wish to undergo regular testing, offer PSA testing every two years from age 50 to age 69, and offer further investigation if the PSA is greater than 3.0 ng/mL.
- In asymptomatic men interested in undergoing testing for early diagnosis of prostate cancer, digital rectal examination is not recommended as a routine test in the primary care setting.
- Do not offer PSA testing to a man who is unlikely to live another seven years.
- Offer active surveillance to men with prostate cancer who meet all the following criteria:
- Advise men with potentially curable prostate cancer considering watchful waiting that their risk of developing more advanced prostate cancer and dying from it will be higher with watchful waiting than with immediate definitive treatment but that, in the medium to long term, watchful waiting is unlikely to diminish their wellbeing and quality of life.
Offer evidence-based decisional support to men considering whether or not to have a PSA test, including the opportunity to discuss the potential benefits and risks of PSA testing before the decision to test is confirmed.
Active surveillance and watchful waiting
Advertisement
PSA ≤ 20 ng/mL, clinical stage T1-2 and Gleason score 6.
Guidelines development panel member and leading cancer epidemiologist Emeritus Professor Bruce Armstrong AM, University of Sydney, noted that the draft guidelines did not recommend a national screening program, in keeping with health policy worldwide. “The draft guidelines are intended to inform the interactions between men and their doctors, when a man might request a PSA test or his doctor might offer one”,said Professor Armstrong AM.
Advertisement
Associate Professor Anthony Lowe, PCFA CEO comments: “Crucial to the development of these new draft guidelines is that a wide variety of stakeholders worked collaboratively to gain consensus. It means we are able to make the most informed recommendations to date on the benefits and harms of PSA testing, and the optimum time to initiate treatment to save lives.”
Following today’s launch as part of the opening of the 2014 World Cancer Congress, health professionals and interested members of the public from around the world are invited to review and comment on the draft guidelines at: wiki.cancer.org.au/australia/Guidelines:PSATesting. Following the consultation phase, the draft guidelines will be submitted to the National Health and Medical Research Council of Australia for approval.
Source-Medindia


![Prostate Specific Antigen [PSA] & Prostate Cancer Diagnosis Prostate Specific Antigen [PSA] & Prostate Cancer Diagnosis](https://www.medindia.net/images/common/patientinfo/120_100/prostate-specific-antigen.jpg)