‘Identification of a region of the genome in the kidney helps develop new treatment mechanisms for bone disorders.’
Tweet it Now
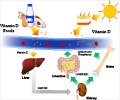
Vitamin D is essential for building bones. Inactive vitamin D obtained from the diet or produced in the skin is converted to its active form, calcitriol, in the kidneys. Calcitriol can also be produced in other parts of the body, including skin cells and immune cells. In these non-kidney sites, calcitriol seems to serve an altogether different purpose: it appears to play no role in regulation of minerals, and its production is induced by inflammation rather than by the hormones that control calcitriol synthesis in the kidneys.
Understanding the function of non-kidney calcitriol production and its connection to inflammatory diseases, ranging from multiple sclerosis to arthritis, is of great interest to a number of biomedical researchers, who would like to know whether and how vitamin D could potentially be used to treat these diseases. However, research in this area has been impeded by the inability to separately study the pathways controlling kidney vs. non-kidney vitamin D activation.
A group of researchers at the University of Wisconsin, overseen by J. Wesley Pike, have now overcome this hurdle by using CRISPR/Cas engineering to produce mice with kidney-specific control of vitamin D activation.
"Through the creation of these mice, we can turn off endocrine regulation of [calcitriol] production exclusively in the kidney," said Mark Meyer, the research associate who led the new study.
Advertisement
Calcitriol production is carried out by the enzyme CYP27B1, but the genomic regions that the team deleted in the experimental mice are far away from the gene encoding this enzyme. Rather, the researchers deleted what's known as enhancer regions for the Cyp27b1 gene.
Advertisement
Importantly, deleting these regions affected the response of vitamin D activation to hormones, but not to inflammation-related molecules, indicating that these regions were key to allowing researchers to separately study the two types of calcitriol production.
While the long-term goal of the team is to understand the function of calcitriol in inflammatory diseases, identifying the kidney-specific enhancer region of the genome is already yielding insights into the mechanisms involved in bone disorders.
Meyer described the work of a clinician collaborator who, in aiming to understand pediatric skeletal disorders, is examining his patients for mutations in the same enhancer regions identified in the mice.
"That [information] might be able to tell that particular clinician about which treatment course might be most appropriate for that individual," Meyer said. For example, many skeletal disorders are treated with calcium and vitamin D supplements, but patients with mutations in regions involved in vitamin D regulation might actually be harmed by calcium supplementation.
Source-Eurekalert