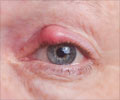
The drug Macugen was approved by the Food and Drug Administration (FDA) to treat age-related vision loss known as macular degeneration was reported to show serious allergic reactions. Pfizer Inc. and OSI Pharmaceuticals said that with use of their eye drug Macugen along with other medications resulted in allergic reactions. FDA says that the companies have already notified the doctors about the reactions, which included allergic swelling around the eyes or lips and other anaphylactic symptoms. After the complaints the companies have added the information about these in their drug's label.
Due to this Pfizer shares closed down 1.6 % at $24.69 on the New York Stock Exchange. Shares of OSI at 3.5 % at $30.02 on the Nasdaq. The companies also warned doctors to evaluate patients for a history of hypersensitivity reactions before giving the drug. The drug makers said that the complaints came in after the injectable drug was approved in the United States in 2004. The sensitivity occurred when Macugen (pegaptanib) was given along with other medications as part of the injection procedure.Eye Drug Macugen Shows Allergic Reactions
Recommended Readings
Advertisement