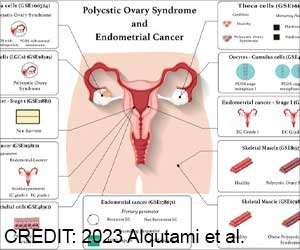
Zolinza, the most recent skin cancer drug has today received approval from the U.S. Food and Drug Administration (FDA). Zolinza (vorinostat) capsules is mainly recommended for the treatment of cutaneous T-cell lymphoma (CTCL) type of skin cancer when the disease persists, worsens, or returns during or after treatment with other medicines.
Zolinza has been approved as part of FDA's Orphan Drug program that offers companies financial incentives to develop medications for diseases which affect less than 200,000 American patients a year. About three in every one million people are diagnosed with CTCL each year in the United States. Most of those afflicted with CTCL are men with an average age of 50 years.Steven Galson, M.D., director of FDA's Center for Drug Evaluation and Research said, "This approval is another example of the benefits of modern research that's focused on providing prescribers with safe and effective therapies for all types of cancer, including those that affect relatively few patients."
Two clinical trials that were conducted with 107 CTCL patients gave evidence of Zolinza's safety and effectiveness. These patients were treated with Zolinza following return of their disease following other treatments.
30 percent of patients who received Zolinza observed an improvement in a scale that was defined by scoring of skin lesions and lasted an average of 168 days.
Some of the most common serious adverse events that occurred were pulmonary embolism, deep vein thrombosis, dehydration, and anemia. Some of most common other adverse events were gastrointestinal symptoms such as nausea, diarrhea, vomiting, anorexia and constipation; fatigue; chills; and taste disorders.
However Zolinza has shown probable fetal harm if administered during pregnancy according to results of animal studies.
Advertisement
Source-Medindia
NLA