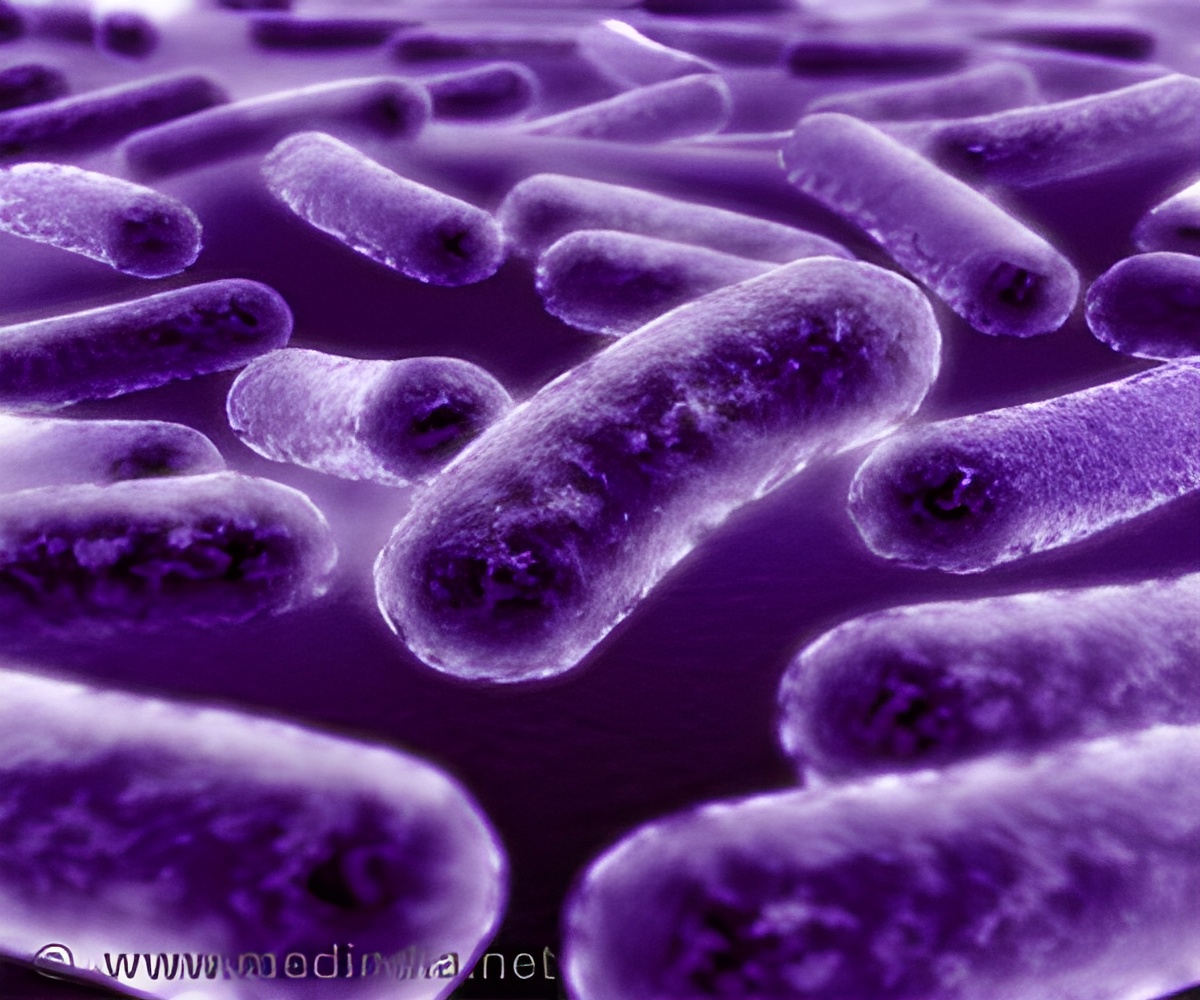
‘Chitin receptors commonly found in shells of crabs can signal Vibrio cholerae bacteria to produce behaviors very different from those seen in human cholera disease.’
Tweet it Now
Among the cholera strains studied, less than a quarter were able to take up DNA from other sources. Almost all of the samples taken from the environment were able to kill other bacteria - a phenomenon called "bacterial dueling" - but just 14 percent of the bacterial pathogen strains isolated from humans had that capability. "It's a dog-eat-dog world out there even for bacteria," said Brian Hammer, an associate professor in the School of Biology at the Georgia Institute of Technology. "Bacteria such as Vibrio cholerae sense and respond to their surroundings, and they use that information to turn on and off the genes that benefit them in the specific environments in which they find themselves."
The research, supported by the National Science Foundation and the Gordon and Betty Moore Foundation, provides information that could lead to development of better therapeutic agents against the disease, which is found in densely-populated areas with limited sanitation and clean water. The research was done with assistance from the Centers for Disease Control and Prevention (CDC), and was reported in the journal Applied and Environmental Microbiology.
In humans, the cholera bacteria produce a diarrheal disease that can kill untreated patients in just a few hours. The deadly effects of the disease, however, are actually caused by a virus that infects the Vibrio cholerae strains found in humans. The toxin carried by the virus helps spread the disease among humans, but cholera strains quickly lose the virus and adapt other competitive mechanisms in the environment.
To study how cholera regulates these adaptations, Georgia Tech graduate student Eryn Bernardy obtained nearly 100 samples of cholera bacteria from a variety of sources globally, including one originally isolated from a 1910 Saudi Arabian outbreak of the disease. She then studied 53 of the samples for their ability to (1) degrade chitin, (2) take up DNA from the environment, and (3) kill other bacteria by poking them with a poisoned spear.
Advertisement
To study their ability to take up DNA, bacterial cells were grown on crab shells, then exposed to raw DNA containing a gene for antibiotic resistance. The cells were scraped off the shells and placed onto agar plates containing an antibiotic that would normally kill the bacteria. Colonies that survived showed they had taken up the genetic material.
Advertisement
"We found a very sharp difference between the clinical isolates and the environmental isolates," Hammer said. "For example, most of the isolates that came out of patients either couldn't kill other bacteria, or were carefully controlling that behavior. Patient isolates have a very different way of competing inside the human body. They use the virus-encoded toxin to cause the diarrheal disease and remove their competitors from the intestine."
With help from CDC scientists, the researchers correlated the behavior of each strain with their unique DNA sequences. They also examined the strains for the presence of the toxin used to cause disease.
To deduce the rules governing the bacterium's behavior, Hammer and his lab have been studying cholera for the last 15 years, starting with a single strain first isolated in Peru in the early 1990s. When a cholera outbreak began in Haiti after the 2010 earthquake, his lab worked with the CDC to isolate these new strains. In further study, Hammer was surprised to find that the 2010 Haitian strains were less capable than the 1991 Peruvian variety.
"We were very surprised to find that most of the Haiti strains did not behave like the one we had been studying for years," he said. "This was a reminder to us that we needed to embrace the diversity of the organisms we've been studying. We thought this would be an opportune time to start looking at how diverse Vibrio cholerae really is."
Hammer compared the diversity of the cholera strains to the diversity of humans, who increasingly receive personalized health care.
"In humans, one size doesn't fit all for patient care," he said. "For cholera, the behavior is personalized for each strain. Understanding this will be useful in the development of future therapeutics, and we're hopeful that knowing how these bacteria interact with other organisms in complex communities will lead us to things that can truly benefit humans."
Source-Eurekalert