A team at the Spanish National Cancer Research Centre (CNIO) led by María Blasco has successfully created a transgenic mouse model that simulates the disease in humans. And its study has allowed them to demonstrate the process linking telomere impairment with the condition. Their results are published today in the online edition of the journal Blood.
Telomeres and stem cells
Telomeres consist of a repetitive DNA sequence bound to a series of proteins, including Trf1, which guard them from degradation and/or damage. Using transgenic techniques, Blasco's team have managed to eliminate the Trf1 protein from mouse bone marrow, in order to explore its role in the tissue's function.
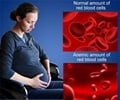
They found that when Trf1 is eliminated, the mice develop exactly the same symptoms as aplastic anaemia sufferers: bone marrow failure with the corresponding pancytopenia (a reduction in the number of red and white blood cells, as well as platelets). Also, the authors have shown for the first time that the absence of this protein causes a shortening of the telomeres of blood cell-producing stem cells which leads, in turn, to the progressive stress-induced death of the remaining stem cells in the tissue and, eventually, the death of the animal.
This discovery establishes the molecular bases of certain genetic variants of aplastic anaemia and opens a new line of attack via Trf1 to prevent the telomere shortening and cell death that trigger the disease. "We have generated an animal model for aplastic anaemia associated with short telomeres that may aid in the design and testing of new therapeutic strategies," confirms Blasco. These findings may also offer insights into other processes linked to telomere length, such as ageing and cancer.
Advertisement
Source-Eurekalert