‘Tumor cells collected during the removal of a cancerous bladder and - in some cases - transplanted into mice with weakened immune systems, could help physicians rapidly identify high-risk cancers.’
Tweet it Now
The researchers, based at the Ludwig Center at the University of
Chicago, found that detection of poorly differentiated basal tumor cells
in early stage cancers; overexpression in those cells of a gene known
as cell division cycle 25C, which plays a key role in the regulation of
cell division; or the ability of tumor fragments to grow when
transplanted into a mouse, all predicted an increased risk of death. "If confirmed in larger studies, our findings could help physicians get a better handle on how a patient's bladder cancer is likely to progress and allow them to personalize treatment based on that knowledge," said study author Ralph Weichselbaum, the Daniel K. Ludwig Distinguished Service Professor of Radiation and Cellular Oncology and Chair of the Department of Radiation and Cellular Oncology at the University.
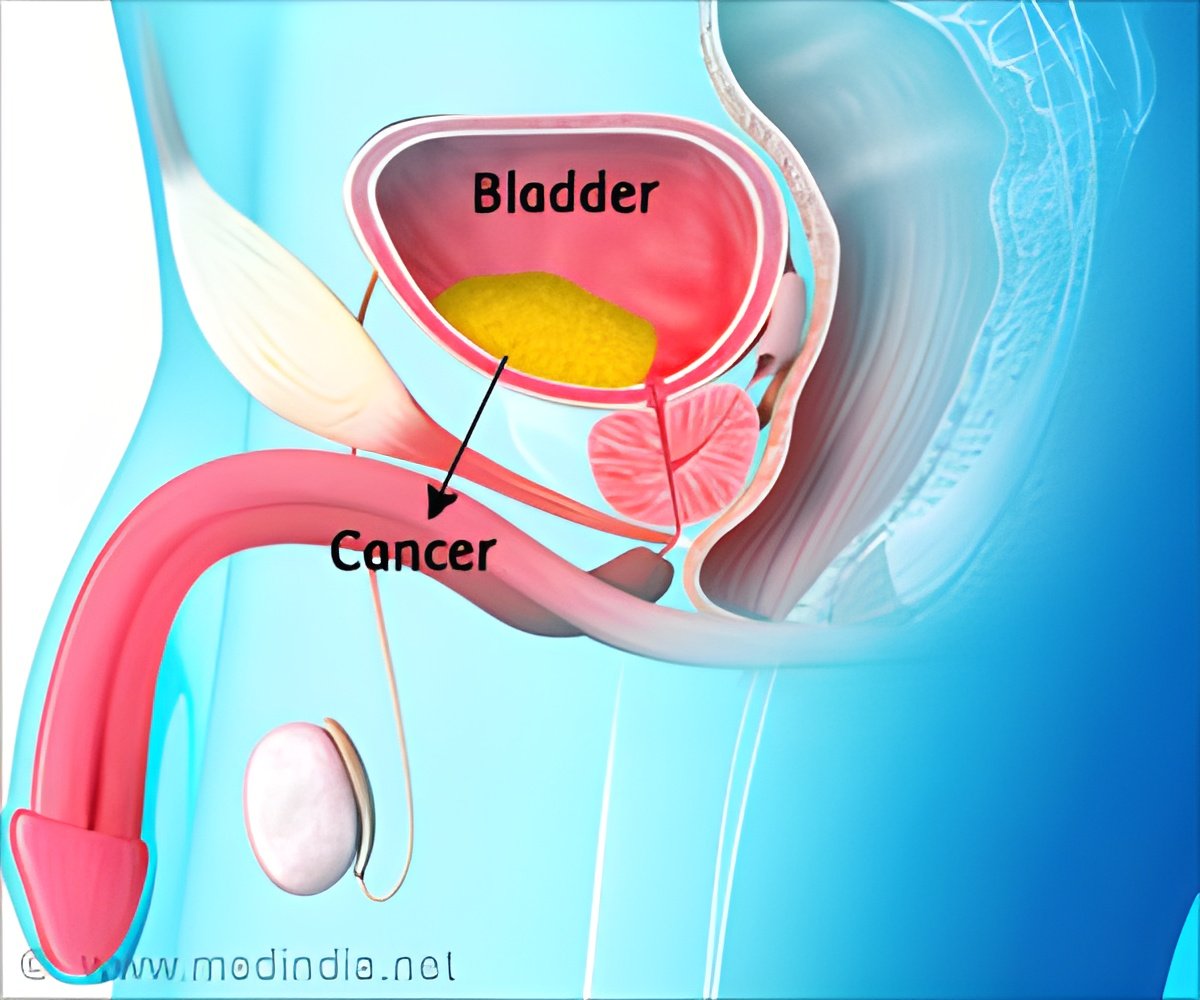
Weichselbaum and colleagues obtained tumor samples from 71 bladder cancer patients treated at the University and used flow cytometry to isolate and count specific subtypes of tumor cells in each sample. They showed that an excess of one relatively rare subtype in early-stage cancers - the basal tumor cell (BTC) - was associated with a three-fold increase in risk of death.
Analyzing the global expression of genes in BTCs, the researchers also identified a potentially prognostic biomarker for bladder cancer: cell division cycle 25C (CDC25C), a protein that drives cell division. An expanded analysis, including 400 bladder cancer patients, found that the expression of this protein is associated with an increased risk of death even after the removal of the cancerous bladder.
This association disappeared in patients who had previously received chemotherapy. A test for CDC25C could, the authors suggest, help determine whether a bladder cancer patient is likely to benefit from drug treatment.
Advertisement
"Prognostic knowledge can change a lot about how you choose to treat a cancer," said Weichselbaum. "We may be able to avoid aggressive measures if we find a tumor has relatively few basal cells," he said. "We could treat an early-stage bladder cancer with less aggressive therapy, avoiding debilitating interventions like radical cystectomy. But if a bladder tumor has a lot of basal cells, we may need to take the entire bladder out and follow that with chemotherapy.
Advertisement
Source-Eurekalert