One-year survival rates were 35% and 22%, respectively, and two-year survival rates were 9% and 4%, respectively. Similar side effects were found in the new drug and gemcitabine alike. The trial report therefore concluded that the nab-paclitaxel/gemcitabine combination significantly improves overall survival and response rate in patients with advanced pancreatic cancer.
Dr Josep Tabernero, Head of the Medical Oncology Department at the Vall d´Hebron University Hospital, Director of the Vall d´Hebron Institute of Oncology (VHIO), and one of the co-authors of the study, confirmed that "the results of this study take a step in the right direction towards controlling advanced pancreatic cancer, a disease with a very poor prognosis. We are confident that the gemcitabine/nab-paclitaxel combination will become one of the reference treatments for this disease. New drugs in the pipeline in this field are already being gauged against this combination as the benchmark to beat. Along these lines, investigators at VHIO are developing promising laboratory model combinations for pancreatic cancer treatment."
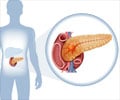
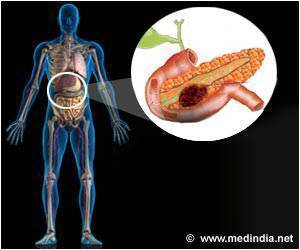
New first-line treatment for pancreatic cancer
Pancreatic cancer is the fourth leading cause of cancer-related death in Europe and the United States. Gemcitabine therapy has been the standard first-line treatment in patients with unresectable, locally advanced or metastatic pancreatic cancer since 1997. In metastatic disease, one-year survival ranges from about 17 to 23%, while five-year survival is only 2%.
Although numerous phase II and III studies have been conducted on new drugs combined with gemcitabine, most have not shown significant improvement in survival among these patients. The nab-paclitaxel/gemcitabine combination opens a new treatment avenue in this patient population.
Advertisement