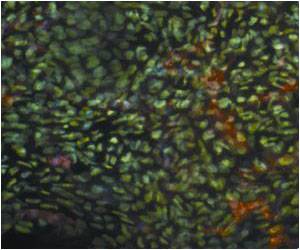
In a study led by Douglas Hanahan, researcher at EPFL and holder of the Merck Serono Chair in Oncology, scientists sought to examine the role of copper in cancer. To do this, they used genetically engineered mice with pancreatic neuroendocrine tumors. "This study was motivated by our previous puzzling observation; namely that cancers, unlike healthy tissues, are especially sensitive to changes in systemic copper levels", said Seiko Ishida, the lead author of the paper.
Their research provides direct evidence that copper can enhance the proliferation of cancer cells. "The biggest surprise was that a small amount of copper added to drinking water accelerated the growth of tumors, indicating that copper is an essential nutrient for them, said Ishida."
Teaming up with Johan Auwerx, also at EPFL, the researchers found that copper insufficiency resulted in a lower activity of the respiration enzyme in tumors. PET scans also revealed that copper-deficient tumors took higher levels of glucose, suggesting that their cells were compensating more and more by using glycolysis rather than respiration for their energy. But despite this, ATP levels did not fully recover, and tumors did not grow further.
Importantly, the researchers do not think that copper causes cancer. Exposure of healthy mice to the same amount of copper via drinking water for up to two years did not result in an increased incidence of cancer. The authors suggest that copper levels could be monitored in cancer patients. They propose that minimizing copper in the patient's system may be beneficial in cancer therapy, especially when combined with drugs that block glycolysis. This two-step strategy would starve cancer cells – which tend to require much higher amounts of energy than normal cells – by limiting their two major pathways for ATP production.
Source-Eurekalert