Kaufmann emphasized that vaccines can play an important role in turning the tide. He referred to studies showing that a 40-50 percent reduction in TB could be achieved by new vaccines. Improved drugs could lead to 10-27 percent reduction and better diagnostics to a 13-42 percent drop.
He elaborated on the development of the VPM1002 vaccine candidate by his team. VPM has successfully completed tests on safety and immunogenicity in Germany. Further trials (phase Ib) are taking place in South Africa at the moment.
Also in Spain progress is made. "It is a difficult process from research to development," Professor Carlos Martin of the University of Zaragoza said, describing the development of the MTBVAC01 vaccine candidate discovered by him and his colleagues. After more than 10 years of discovery and proof of concept and now 4 years of development, the vaccine is taking its first steps out of the lab: The vaccine is scheduled to be tested in people (phase I safety trials) the end of next year.
Dr.Jelle Thole, director of TuBerculosis Vaccine Initiative (TBVI), in a meeting with Spanish journalists called MTBVAC01 a leading vaccine in its kind. "It is the only candidate derived from the actual bug that starts tuberculosis. All other vaccine candidates either are based on improving or boosting BCG," he explained. TBVI, a European research consortium for the development of new TB vaccines, aims to have 8 vaccines in phase II safety and efficacy trials in 10 years.
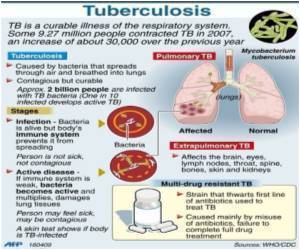
New vaccines are aimed to improve or replace BCG, the only currently available vaccine against tuberculosis, which "does not protect against the most prevalent form of the disease, and therefore has little - if any - impact on the epidemiology of TB," Kaufmann said.
Advertisement
Worldwide new tools against tuberculosis are in several stages of development. Jan Gheuens, a senior program officer on the tuberculosis team at the Bill & Melinda Gates Foundation, summarized at the symposium: "Two vaccines are in large clinical trials, lots of vaccines are in other phases. Furthermore, 8 drugs are in pre-clinical development or further. And there is much excitement on the performance of a TB molecular diagnostic test."
Advertisement
The two day symposium (3-4 June 2010) was organized by the University of Zaragoza, the foundation Ramon Areces and TuBerculosis Vaccine Initiative (TBVI).
Contributed by: Bobby Ramakant
Source-Medindia