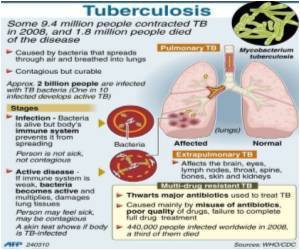
TB remains one of the major causes of disability and death worldwide, with an estimated 1.8 million deaths in 2008 and an increasing incidence of drug-resistant disease. New, less costly tools to rapidly diagnose TB and its drug-resistant forms are urgently needed to help stem the tide of new infections.
The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, has supported the development of this diagnostics technology since 2002. This support was critical to the public-private partnership that led to the Xpert MTB/RIF TB test. The current clinical trial findings are from a study conducted by the Foundation for Innovative New Diagnostics (FIND), a Swiss-based nonprofit organization. NIAID currently supports a partnership project to expand the diagnostic system to detect other resistance markers for extensively drug-resistant TB.
Source-Eurekalert












