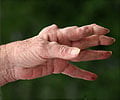
Proven combinations of medicines and the introduction of new anti-arthritis drugs have significantly improved the treatment of rheumatoid arthritis (RA), according to guidelines issued by the American College of Rheumatology.
Lead author Kenneth Saag, M.D., M.Sc., a professor in the UAB Division of Clinical Immunology and Rheumatology, said the new guidelines update strategies for treating RA with the goal of preventing joint damage and disability.The new recommendations do not strive to replace individualized medical decisions, Saag said. Instead, they are meant to guide rheumatologists and other health care workers toward the most updated recommendations. The last set of American College of Rheumatology RA treatment guidelines was published in 2002.
“The recommendations developed are not intended to be used in a ‘cookbook’ or prescriptive manner, or to limit a physician’s clinical judgment,” Saag said. “They provide guidance based on clinical evidence and expert panel input.”
The recommendations focus on several classes of anti-arthritic drugs, including a potent group of agents called disease-modifying anti-rheumatic drugs (DMARDs). Newer genetically engineered DMARDs called biologics often work in combination with earlier therapies. Many anti-arthritic drugs are designed to stop damaging inflammation, and biologics work to interrupt the chain of events that leads to inflammation.
Newer biologics called anti-TNF agents – adalimumab (Humira), etanercept (Enbrel) and infliximab (Remicade) – prevent the production of an immunity protein that plays a role in inflammation.
Some of the key recommendations include:
Advertisement
- Methotrexate or leflunomide therapy is recommended for most RA patients.
- Anti-TNF agents etanercept, infliximab, or adalimumab along with methotrexate can be used in new or early RA cases with worsening and severe symptoms.
- Doctors should not initiate or resume treatment with methotrexate, leflunomide, or biologics if RA patients have active bacterial infection, shingles (herpes-zoster), hepatitis B, hepatitis C and active or latent tuberculosis.
Source-Newswise
SRM