In the study, a sample of over 9,000 patients who received the latest stents were no more likely to die in the few years following the procedure, compared to a matched sample of over 9,000 patients who underwent bypass surgery instead.
Clinical practice in this area is still largely guided by past studies of bare-metal and first-generation drug-coated stents—studies that found greater long-term risks of heart attack and death from the devices, compared to traditional bypass surgery.
“We aren’t using older stents anymore, so this study gives us some insight into more up-to-date clinical outcomes,” says Sripal Bangalore MD, an associate professor in NYU Langone’s Leon H. Charney Division of Cardiology, Department of Medicine, who was lead author of the study.
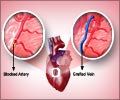
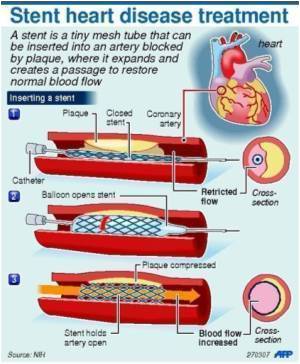
Stents are metal mesh tubes that are threaded into the heart through a leg or an arm artery using a catheter. They are then expanded to keep a blocked coronary artery open indefinitely. Introduced in the late 1980s, they are now commonly used to open blocked arteries in acute heart attack cases, and to widen narrowed vessels in less severe cases of cardiovascular disease. Hundreds of thousands of patients receive stents each year in the U.S.
The technique for inserting a stent—called percutaneous coronary intervention, or PCI—requires a much shorter hospital stay than traditional open-heart bypass surgery, and is much less likely to trigger strokes.
Advertisement
The new study made use of data from New York state registries that track cardiovascular procedures. From nearly 35,000 cases during 2008-2011, the researchers selected two groups of 9,233 patients with multi-vessel disease that were closely matched in age and other relevant measures. One group had undergone bypass surgery, the other PCI with a new generation of stent that is coated with the drug everolimus to inhibit inflammation and scar tissue growth.
Advertisement
Patients in the stent group got about 1.5 times the number of heart attacks long-term, compared to the bypass group, but for those patients whose stents successfully opened all of their diseased arteries, there was no significant increase in heart attacks.
Bypass surgery showed a clear advantage only in the percentages of patients who had to return to have an artery reopened: 3.1 percent for the bypass group, versus 7.2 percent per year for the stent group.
“The key finding is that there was no greater mortality in the sample of patients receiving the latest generation stents,” says Dr. Bangalore. “This may encourage patients and physicians to weigh the risks and benefits of stenting versus bypass surgery, now that we have further information.”
Although this was an observational study, not a randomized clinical trial, Dr. Bangalore notes that its results are consistent with a general trend in New York clinical registry studies. “Over the last twenty years of these registry studies, the gap in apparent mortality risk between bypass and PCI has been getting smaller and smaller as stent technologies have advanced,” he says.
The study was a collaboration between Dr. Bangalore’s group and a team led by Edward L. Hannan, Ph.D., Distinguished Professor of Health Policy, Management and Behavior at the School of Public Health, SUNY-Albany. Besides Dr. Bangalore and Dr. Hannan, co-authors were Yu Guo, Saul Blecker, MD and Jinfeng Xu, PhD of NYU Langone, and Zaza Samadashvili, MD of the School of Public Health at SUNY-Albany.
The study was funded by Abbott Vascular, a division of Abbott Laboratories, which makes one of the two everolimus-coated stents now on the market.
Source-Medindia