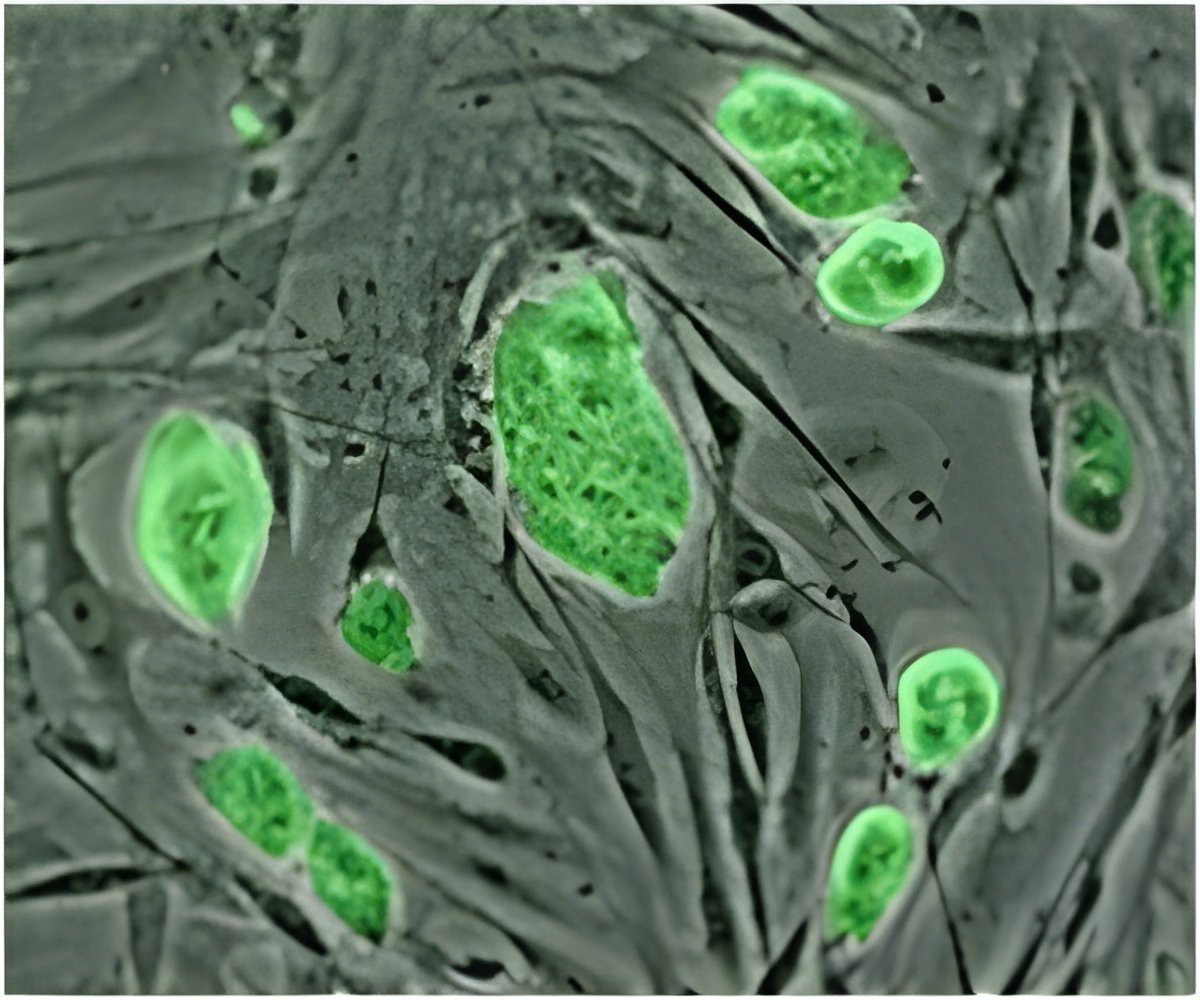
In a study published in the June 5 online edition of the journal Science Signaling, the team reported that tinkering with a protein molecule called p190RhoGAP shaped the development of cardiac stem cells, prodding them to become the building blocks for either blood vessels or heart muscle. The team members said that by altering levels of this protein, they were able to affect the future of these stem cells.
"In biology, finding a central regulator like this is like finding a pot of gold," said Andre Levchenko, a biomedical engineering professor and member of the Johns Hopkins Institute for Cell Engineering, who supervised the research effort.
The lead author of the journal article, Kshitiz, a postdoctoral fellow who uses only his first name, said, "Our findings greatly enhance our understanding of stem cell biology and suggest innovative new ways to control the behavior of cardiac stem cells before and after they are transplanted into a patient. This discovery could significantly change the way stem cell therapy is administered in heart patients."
Earlier this year, a medical team at Cedars-Sinai Medical Center in Los Angeles reported initial success in reducing scar tissue in heart attack patients after harvesting some of the patient's own cardiac stem cells, growing more of these cells in a lab and transfusing them back into the patient.
Using the stem cells from the patient's own heart prevented the rejection problems that often occur when tissue is transplanted from another person.
Advertisement
During their research, the Johns Hopkins team members wondered whether changing the surface where the harvested stem cells grew would affect the cells' development. The researchers were surprised to find that growing the cells on a surface whose rigidity resembled that of heart tissue caused the stem cells to grow faster and to form blood vessels. A cell population boom occurred far less often in the stem cells grown in the glass or plastic dishes typically used in biology labs. This result also suggested why formation of cardiac scar tissue, a structure with very different rigidity, can inhibit stem cells naturally residing there from regenerating the heart.
Advertisement
"It was the kind of master regulator of this process," Levchenko said. "And an even bigger surprise was that if we directly forced this molecule to disappear, we no longer needed the special heart-matched surfaces. When the master regulator was missing, the stem cells started to form blood vessels, even on glass."
A final surprise occurred when the team decided to increase the presence of p190RhoGAP, instead of making it disappear.
"The stem cells started to turn into cardiac muscle tissue, instead of blood vessels," Levchenko said. "This told us that this amazing molecule was the master regulator not only of the blood vessel development, but that it also determined whether cardiac muscles and blood vessels would develop from the same cells, even though these types of tissue are quite different."
But would these lab discoveries make a difference in the treatment of living beings? To find out, the researchers, working on the heart-matching surfaces they had designed, limited the production of p190RhoGAP within the heart cells. The cells that possessed less of this protein integrated more smoothly into an animal's blood vessel networks in the aftermath of a heart attack. In addition, more of these transplanted heart cells survived, compared to what had occurred in earlier cell-growing procedures.
Kshitiz said that the special heart-like surface on which the cardiac stem cells were grown triggers regulation of the master molecule, which then steers the next steps.
"This single protein can control the cells' shape, how fast they divide, how they become blood vessel cells and how they start to form a blood vessel network," he said. "How it performed all of these myriad tasks that require hundreds of other proteins to act in a complex interplay was an interesting mystery to address, and one that rarely occurs in biology. It was like a molecular symphony being played in time, with each beat placed right at the moment before another melody has to start."
Source-Eurekalert